Invasive aspergillosis (IA) is a life-threatening Invasive Fungal Disease (IFD) affecting immunocompromised patients.
Aspergillus spores are common throughout the environment and with the capacity to invade via the lung, cause a spectrum of disease. IA is difficult to diagnose, as symptoms are non-specific and the sensitivity of traditional techniques – such as respiratory culture – is poor and slow. Delayed diagnosis leads to an increased morbidity and mortality rate, highlighting the urgent requirement for a rapid, sensitive diagnostic method.
Current clinical practice
Due to the constraints of current diagnostic strategies, clinicians rely on prophylaxis and empiric antifungal therapy to manage patients at risk of, or with suspected, IA. Prophylaxis is used as a preventative measure for high risk, non-symptomatic patients, while empirical treatment is given to patients based on non-specific clinical signs of infection (i.e. antibiotic refractory fever). Both methods are costly, and broad use of antifungal drugs may lead to side-effects.
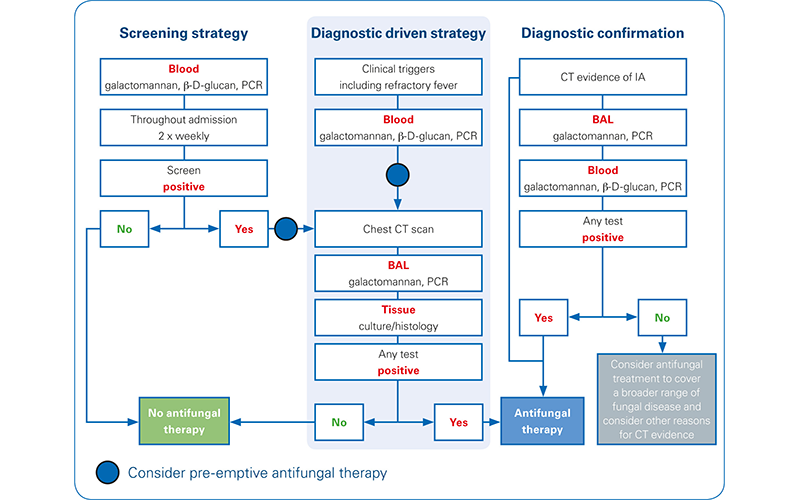
Polymerase chain reaction (PCR) assays (such as Bruker’s Fungiplex® Aspergillus IVD PCR), which target pathogenic DNA, can be used in the systematic testing of high-risk patients to support early detection of invasive aspergillosis (see Figure 1). By including PCR in surveillance biomarker testing strategies, patients can be monitored for the early signs of infection; minimizing the use of empiric therapy and reducing the cost of care with no adverse effect on patient outcomes.
Overcoming the barriers to change
The widespread adoption of Aspergillus PCR into clinical practice has previously been hindered by a lack of standardisation and commercially available tests. Clinicians are often concerned by the risk of a false negative result from a diagnostic test, and this has led to over-prescribing of antifungal drugs. However, results from a specialist laboratory in Wales support the use of systematic PCR testing for the management of patients at-risk of IA. The high sensitivity of PCR, in combination with other biomarkers, allows negative test results to rule out IA diagnosis – thereby enabling a reduction in antifungal treatment, which patients may have been receiving unnecessarily.
The Regional Mycology Reference Laboratory, Public Health Wales, Cardiff, conducted a study assessing the impact of using an Aspergillus PCR and antigen test in a diagnostic algorithm to drive pre-emptive, rather than empirical, antifungal treatment [2]. The results showed a reduction in overt invasive fungal disease by 75% as a result of using pre-emptive treatment. The laboratory was also involved in the validation of the Bruker Fungiplex Aspergillus real-time PCR assay, which produces results in less than two hours following DNA extraction, with a clinical sensitivity of 96% (respiratory culture sensitivity for Aspergillus has been reported at 20-50%) and specificity of 90%.
PCR as the new standard
The commercial availability of a rapid, highly sensitive and selective Aspergillus PCR assay can aid the quicker diagnosis of IA which will significantly improve patient outcomes. In addition to fast detection of Aspergillus DNA direct from patient samples, the adoption of PCR as part of a diagnostic strategy allows clinicians to identify sooner when to start, or stop, prescribing antifungal medication.
For information on Bruker’s Fungiplex® Aspergillus IVD PCR, please click here.
Authors
Dr Jennifer Dougan is a PhD qualified chemist from Bruker Daltonics and Dr Lewis White is the Scientific Head of the UK Clinical Mycology Network Regional Mycology Reference Laboratory
References
[1] A. Mahdi, M. Edwards, R. Barnes, W. Ingram and J. Kell (2014) Investigation and Management of Invasive Fungal Infection (IFI) in Haematological Patients, Cardiff and Vale Health Board Guidance, Cardiff and Vale Trust.
[2] Barnes RA et al. 2009 Clinical impact of enhanced diagnosis of invasive fungal disease in high-risk haematology and stem cell transplant patients. J. Clin. Pathol.62: 64-60. doi: 10.1136/jcp.2008.058354

For more than 55 years, Bruker has enabled scientists to make breakthrough discoveries and develop new applications that improve the quality of human life. Bruker’s high-performance scientific research instruments and high-value analytical solutions enable scientists to explore life and materials at molecular, cellular and microscopic levels.
In close cooperation with our customers, Bruker is enabling innovation, productivity and customer success in life science molecular research, in applied and pharma applications, in microscopy, nano-analysis and industrial applications, as well as in cell biology, preclinical imaging, clinical research, microbiology and molecular diagnostics. For more information, please visit: www.bruker.com.
