A hundred years after Spanish influenza, which is thought to have killed more than 40 million, biomedical scientist and university lecturer Sarah Pitt looks back at the pandemic.
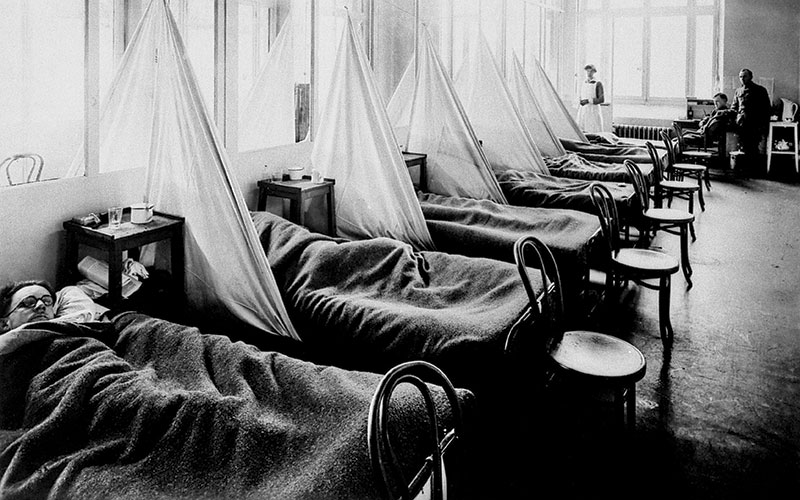
The first influenza pandemic of the 20th century is considered to have started in the Spring of 1918. It continued to cause morbidity and mortality around the world until the end of 1920. The virus affected approximately 1% of the global population and at least 40 million people died. Cases were recorded throughout the world, except in Western Samoa, which had a strict quarantine policy (Oxford et al., 2005). The death toll was therefore considerably higher than the 18 million deaths recorded for military personnel and civilians because of World War I (WWI).
Pandemic influenzas are usually named after the region where they were presumed to have started (see Table 1). The 1918-20 disease was called “Spanish flu”. Although early cases were recorded in many parts of Europe and also the US in 1918, reporting was circumspect due to the ongoing military conflict. Spain, however, was neutral during WWI and so when the King of Spain was affected by influenza it was publicised in the national and international news (Oxford, 2000). This is how the misconception that the pandemic started in Spain and that the index case was a mysterious “Spanish lady” arose.
Influenza A virus
There are four known types of Influenza (A, B, C, D), although only Influenza A and Influenza B are associated with disease in humans. Seasonal influenza, which occurs each winter, involves several co-circulating strains of type A and B viruses that have arisen gradually through antigenic drift (see Text Box 1). Pandemic influenza occurs when genetic re-assortment leads to a new, unexpected Influenza A via antigenic shift (see Text Box 1). Pandemic viruses have different combinations of haemagglutinin (H) and neuraminidase (N) (see Table 1) and they emerge at unpredictable time intervals and in various geographical locations. Many virologists and epidemiologists expected that an influenza pandemic in the late 20th or early 21st century would be as a result of reassortment involving avian and human types of Influenza A and originating in South East Asia. When the 2009-10 pandemic began in South America and genetic analysis implicated swine influenza viruses as well as those from birds and people, it was rather surprising.
The other point to note from Table 1 is that the populations at greatest risk of serious disease and death vary between pandemics. In seasonal influenza, potentially fatal complications and secondary infections are more likely in the very young, very old, or people with underlying illnesses. This was also the case in the Asian and Hong Kong influenza epidemics of the 20th century, However, the Spanish flu (1918-20), Russian flu (1977-78) and Mexican flu (2009-10) pandemics disproportionately affected young adults. The reasons for this are not yet fully understood, but it has been suggested that it is at least partly attributable to immunological factors (McAuley et al., 2015). It is also notable that H1N1 virus types were involved in each case.
Origin of the 1918 pandemic virus
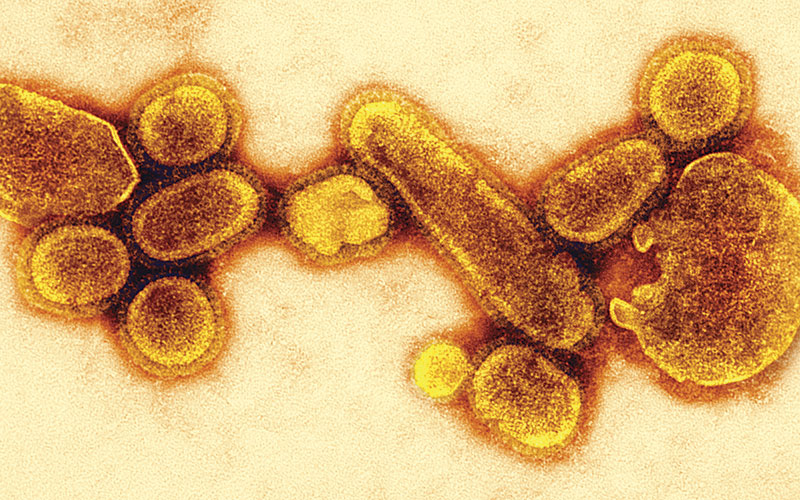
Several possibilities have been proposed for the origin of the 1918 pandemic virus. Serological investigations of survivors of Spanish flu established that the virus was an Influenza A H1N1. More recently, the RNA sequence of influenza virus was identified in fixed and stained histological slides prepared from post-mortem lung tissue collected in 1918 and 1919 from influenza patients (Reid et al., 2003). This confirmed that it was an H1N1 strain, containing genetic elements of human, avian and swine viruses. It is interesting the 2009-10 H1N1 also comprised a combination of human, pig and bird viruses.
There were documented outbreaks of acute respiratory illness which could have been influenza from 1916 onwards in France (on the Western Front), in the UK (Oxford et al., 2002) and China (Humphries, 2014). In the winter of 1916, many soldiers were admitted to the hospital at the British army base in Etaples, Northern France, with symptoms of fever, cough and bronchopneumonia. Mortality rates were up to 50% and post-mortem examination of lung tissue indicated that a “purulent bronchitis” was common. Those who died often developed a characteristic sign of lack of oxygen to the extremities called “heliotrope cyanosis”, named for the pinkish purple colour of the skin. An outbreak of a similar infection was reported from the Aldershot barracks in the UK during March 1917. Retrospective comparison of the symptoms with those seen in the later pandemic indicated that the conditions looked the same clinically. Laboratory tests could not identify any particular bacterium associated with the disease. Experiments conducted with material obtained from patients at Etaples at that time showed that the causative agent was not a bacterium, but a filterable virus (Oxford et al., 2002). This suggests that the pandemic influenza virus emerged in France in 1916 and spread among military personnel through movement of people between the Western Front and the UK. The French army recruited soldiers from their colony of the Annam kingdom (Vietnam, Laos and Cambodia) to fight in the war from 1916. There are reports of outbreaks of a flu-like illness (fever, cough, myalgia) which commonly resulted in complications, described as “Annamite Pneumonia” (which again implies that it was an unfamiliar disease) and pneumococcus infection starting in 1916 (Erkoreka, 2009).
The Western Front in 1916 provided the ideal conditions for an influenza reassortment event. As well as large numbers of people living in basic shelter and overcrowded conditions, soldiers went into local villages to buy live poultry for food and they reared pigs. Thus, there was an opportunity for human, avian and swine strains of influenza to mix. Oxford and colleagues (2002; 2005) suggest that once the emerged H1N1 strain became established in the human population, it continued to cause outbreaks, such as those mentioned above. During this time, the virus mutated by drift (see Text Box 1) to become the virulent form which began to spread around the world in first quarter of 1918. It has been impossible to locate suitable histological samples from any of these pre-1918 outbreaks, which is unfortunate, because the viral sequence would be the best information to test the links between the events.
An alternative theory for the origin of the pandemic virus is that it came from China and was spread to Europe by the workers from the Chinese Labour Corps (Humphries, 2014). These labourers were recruited by the British government and sent to Europe, initially on a route via Singapore and South Africa and then subsequently on a journey which included a stopover in Canada. Thus large numbers of Chinese workers were moved around the world during 1917 and 1918. There are reports of them becoming ill with a strange respiratory illness (which was thought to be plague, but might have been influenza) en route – for example, in Canada in 1917 (Humphries, 2014).
Interestingly, after a short stay in England, these workers were sent to the Western Front, to Etaples. There was an outbreak of “purulent bronchitis” in the Chinese camp in the Spring of 1918. In January of that year, two pigs at that camp died of an infection recorded as “swine fever”. This, therefore, could possibly have been the site of the reassortment event from which the pandemic influenza emerged. However, this theory suggests that the earlier respiratory illness/plague was not the pandemic influenza and lends weight to the idea that it arose in France. Although Oxford et al. (2002) cite accounts from Vera Brittain, who was nursing at Etaples, suggest that Chinese labourers and European soldiers did not mix, it is conceivable that people from the different camps were in contact. However, it is equally likely in that scenario that the virus could have spread from the Europeans to the Chinese.
 The global spread
The global spread

During WW1, large numbers of people were continually moving within and between countries. Not only were soldiers and support workers arriving in Europe from Australasia, South East Asia and North America, but military personnel were going to those countries to help with arrangements. Also volunteers (e.g. nurses) who were too exhausted to work and injured soldiers were moved from the battlefields and in some cases returned to their home countries. There are reports of apparently isolated outbreaks of influenza-like illnesses among soldiers in the US, and the armies on both sides of the conflict in France, Belgium and Eastern Europe in early 1918 (Oxford et al., 2005; Erkoreka, 2009). Although the number of cases was high, most patients recovered. By May, civilians in several countries, including Spain, were being infected and the disease was progressing to the bronchopneumonia with heliotrope cyanosis documented in 1916 and 1917. The incidence of this “Spanish flu” started to increase markedly during the final months of 1918. It spread globally and continued to cause significant morbidity and mortality for the following two years. After the armistice was declared on 11 November 1918, the military camps started to demobilise. Oxford et al. (2005) believe that this was a main contributor to the rapid transmission of the influenza virus at this time, although Humphries (2014) points out that Canadian, US and French armies did not move the bulk of their personnel until well into 1919. However, civilians would certainly have started moving by the end of 1918 and since the war was over, they would have been under less strict control than earlier in the year. It would only have taken a few people incubating the virus arriving in each country to set up the pandemic there.
Antigenic drift & shift
Influenza A is a single stranded negative sense RNA virus. Its genome comprises eight segments.
Antigenic drift
This occurs because transcription errors introduced into the viral genome during RNA replication are not corrected by host mammalian cell mechanisms. Most mutations are detrimental, but occasionally a viable virus arises that competes with other circulating strains of Influenza A.
Antigenic shift
During reassembly after viral replication, one copy of each genome segment must be incorporated into the new virus particle. When a host cell is infected with two or more subtypes of Influenza A, the segments might be a mixture from different viruses. The outcomes of most such “reassortment” events are abortive viruses, but sometimes a transmissible particle occurs.
Reasons for the high mortality
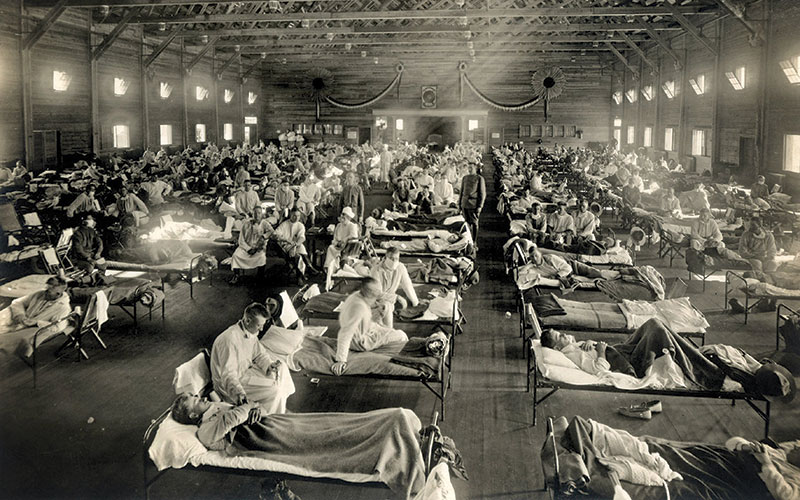

The number of deaths attributed to influenza during the 1918-20 pandemic is more than 10 times that recorded for subsequent pandemics (see Table 1). This could simply be because the virus was extremely widely spread, due to mass movement of people under unusual circumstances. People dispersed from the WW1 battle sites (the presumed site of the reassortment event) to all parts of the world. Public health surveillance and healthcare facilities in many countries would have been compromised due to a focus of personnel and resources on the war effort. There were no antibiotics to treat the secondary bacterial pneumonia infections, let alone intensive care units to support people through respiratory crises. People exposed to the pandemic influenza virus on the WW1 battlefields would have been stressed, exhausted and undernourished. These factors increase susceptibility to an infectious disease. In addition, chemical warfare agents, such as chlorine and mustard gas, were used extensively in the trenches on the Western Front (Oxford et al., 2005). Lung damage caused by these gases may have left soldiers more vulnerable to the complications of a respiratory infection. After several years of the physical and psychological effects of war (displacement, bereavement, lack of food) the civilian population would also have been immunologically vulnerable to influenza. However, it is notable that the mortality rate was also high in Spain (Erkoreka, 2009), which was neutral during WW1
Serious complications and mortality occurred disproportionately among young adults (Taubenberger and Morens, 2006; McAuley et al., 2015). There has been speculation that older adults may have acquired protective antibodies from previous exposure to influenza A viruses, for example, during a pandemic in 1889-90 (Erkoreka, 2009; McAuley et al. 2015). Similarly, younger children infected during an outbreak in 1900 may have developed some immunological resistance to the 1918-20 virus (McAuley et al. 2015). However, the type of influenza A viruses involved in these events cannot be determined, so this remains conjecture. It could simply be that the young adults were more physiologically susceptible. Many young and early middle-aged men would have lung damage from the toxic chemicals and physical injuries as a result of combat. Women of a similar age would have been doing heavier manual labour than previously and with little rest. Prevalence of sub-clinical tuberculosis was probably higher than recognised officially. Another possibility is the nature of the virus itself. The haemagglutinin of influenza A attaches to host respiratory epithelial cells, but there is some variation on the target glycoprotein. Influenza A virus types that affect humans usually attach only to 2-6, sialylated glycan receptors found in lung tissue. Avian viruses have an affinity for 2-3, sialylated glycans, which are also found on human cells, but deeper into the respiratory tract. Analysis of the sequences of viruses isolated from patient samples from the 1918-20 pandemic show a mixture of human and avian types (Taubenberg and Morens, 2006). This was also a feature of the Mexican flu virus, which similarly caused serious pathology and higher mortality than expected in younger adults, particularly pregnant women (Girard et al., 2009). The ability to bind to cells deeper into the respiratory tract than usual could account for the “purulent bronchitis” associated with the 1918-20 H1N1 virus. A possible explanation is that it was caused by an immunological response including inflammation, with strong cytokine and T cell responses (McAuley et al. 2015). The theory is that this effect would have been less marked in children and older people, due to less robust immune systems. In practice, it seems likely that both physiological vulnerability and the virus-induced immunology caused the clinical pathology.
Conclusion
The 1918-20 influenza pandemic is a fascinating topic and some questions may never be answered. From the biomedical scientist’s point of view, it does illustrate the value of collecting, collating and keeping patient samples.
Sarah Pitt is a member of the IBMS Virology Scientific Advisory Panel, Chief Examiner for Virology, an IBMS Fellow and lectures in microbiology. She would like to thank Ms C Dedi and Dr A Gunn for helpful comments on drafts of this article. For the article with full references, visit thebiomedicalscientist.net
References
Erkoreka, A. (2009). Origins of the Spanish Influenza pandemic (1918–1920) and its relation to the First World War. Journal of Molecular and Genetic medicine, 3(2) 190-194.
Girard, M.P., Tam, J.S., Assossou, O.M. and Kieny, M.P., (2010). The 2009 A (H1N1) influenza virus pandemic: A review. Vaccine, 28(31): 4895-4902.
Humphries, M.O., (2014). Paths of infection: The First World War and the origins of the 1918 influenza pandemic. War in History, 21(1): 55-81.
McAuley, J.L., Kedzierska, K., Brown, L.E. and Shanks, G.D. (2015). Host immunological factors enhancing mortality of young adults during the 1918 influenza pandemic. Frontiers in immunology, 6: 419.
Oxford, J.S. (2000). Influenza A pandemics of the 20th century with special reference to 1918: virology, pathology and epidemiology. Reviews in Medical Virology, 10(2): 119-133.
Oxford, J.S., Sefton, A., Jackson, R., Innes, W., Daniels, R.S. and Johnson, N.P.A.S., (2002). World War I may have allowed the emergence of “Spanish” influenza. The Lancet Infectious Diseases, 2(2): 111-114.
Oxford, J.S., Lambkin, R., Sefton, A., Daniels, R., Elliot, A., Brown, R. and Gill, D., (2005). A hypothesis: the conjunction of soldiers, gas, pigs, ducks, geese and horses in Northern France during the Great War provided the conditions for the emergence of the “Spanish” influenza pandemic of 1918–1919. Vaccine, 23(7): 940-945.
Reid, A.H., Janczewski, T.A., Lourens, R.M., Elliot, A.J., Daniels, R.S., Berry, C.L., Oxford, J.S. and Taubenberger, J.K., 2003. 1918 Influenza Pandemic and Highly Conserved Viruses with Two Receptor-Binding Variants. Emerging Infectious Diseases, 9(10): 1249-1253.
Taubenberger, J.K. and Morens, D.M. (2006). 1918 Influenza: the mother of all pandemics. Emerging Infectious Diseases, 12(1):.15- 22.
