Principal Biomedical Scientist Advanced Practitioner in Morphology Nicki Lawrence looks at three case studies of patients who were bitten by dogs and developed a Capnocytophaga canimorsus infection.

Review of a blood film from a patient with sepsis is frequent within morphology. These patients are often critically ill and usually in an intensive care or high-dependency setting. Examination of the blood film provides a wealth of information and in this article we review a cluster of cases where the patient has experienced a dog bite and developed an infection with Capnocytophaga canimorsus.
A rare infection
C. canimorsus is an anaerobic Gram-negative rod that is part of the oral commensal flora of cats and dogs that can be transmitted to humans by penetrating bites. Infection in humans is rare with an infection rate between 0.5 and 0.67 cases per million. C. canimorsus mostly affects males over the age of 50 years. It is most common in immunocompromised patients and patients who are on corticosteroids, have liver cirrhosis or abuse alcohol. These groups of patients comprise 60% of Capnocytophaga infections with the CDC commenting that asplenic patients have a 30–60 times greater risk of death with rapid progression to organ failure and death within 24 to 72 hours. However, a small number of fatal cases have been reported in immunocompetent patients. A recent systematic review showed the case fatality rate of C. canimorsus in immunocompetent patients was 29.7% between 2002 and 2019.
Infection with C. canimorsus can range from a self-limiting disease to severe infection and eventual death in as little as 24 hours, often due to complications from shock, sepsis, disseminated intravascular coagulation (DIC), and organ failure. Signs and symptoms of C. canimorsus infection can occur within three to five days after the animal bite, but can take up to 14 days to appear. Sepsis is the most common manifestation – “a life-threatening organ dysfunction caused by a dysregulated host response to infection”. Septic shock is defined as a subtype of sepsis, with haemodynamic and metabolic abnormalities, and is associated with increased mortality. Other complications include skin infections, cellulitis, myocardial infarction, renal failure and amputation of extremities due to DIC.
Figures 1–5: blood films for the three case study patients
Figure 1 (patient 1): ×600 magnification. Image below shows a heavily vacuolated neutrophil. There are 2 rod-shaped structures present (arrowed).
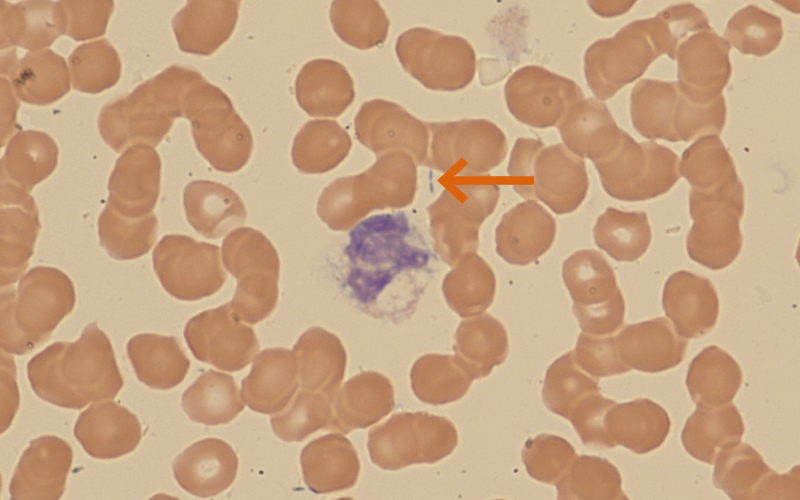
Presentation Patient 1
- Hb 147 g/L (120–170)
- WBC 5.2 x109/L (4.0–11.0)
- Platelets 7 x109/L (150–450)
- Normal WBC differential
- Normal INR and APTT
- D-dimer 63,450 ng/mL FEU (1–499)
- CRP 424 mg/L (0–5)
- LDH 1406 IU/L (115-235)
Figure 2 (patient 2): ×600 magnification. Image shows marked anaemia, with some rouleaux also seen. The neutrophil present has critical blue-green neutrophil inclusions (arrowed).
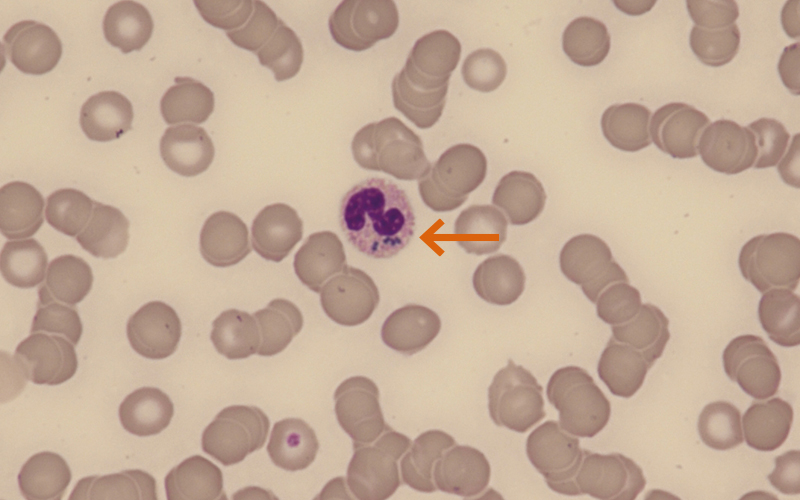
Presentation Patient 2
- Hb 62 g/L (130–180)
- WBC 14.3 x109/L (4.0–11.0)
- Platelets 107 x109/L (150–450)
- Neutrophils 13.6 x109/L (2.0–7.5)
- D-dimer 17,923 ng/mL FEU (1–499)
- Fibrinogen 0.97 g/L (1.9–4.8)
- CRP 427 mg/L (0–5)
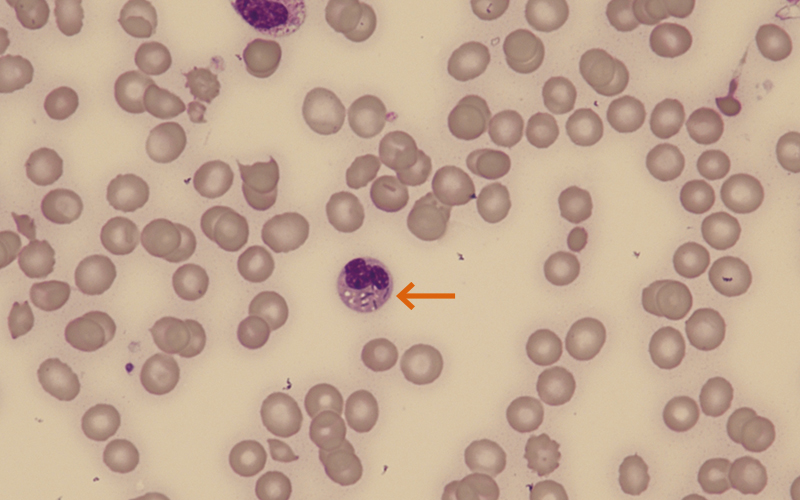
Figure 3 (patient 2): ×600 magnification.
Image shows occasional schistocytes, helmet cells and microspherocytes present. The neutrophil present has numerous rod-like structures within the cytoplasm.
Figure 4 (patient 3): ×600 magnification. Day 3 blood film shows schistocytes are easily seen. Red cell anisocytosis is present and polychromasia can also be seen – reticulocyte count 8.1%.
Patient 3 Day 3
- Hb 121 g/L (130–180)
- WBC 2.9 x109/L (4.0–11.0)
- Platelets 28 x109/L (150–450)
- Neutrophils 2.6 x109/L (2.0–7.5)
- D-dimer 22,202 ng/mL FEU (1–499)
- Fibrinogen 0.43 g/L (1.9–4.8)
- CRP 586 mg/L (0–5)
- LDH 2026 IU/L (115–235)
- ALT 155 IU/L (0–48)
- Bilirubin 54 µmol/L (0–21)
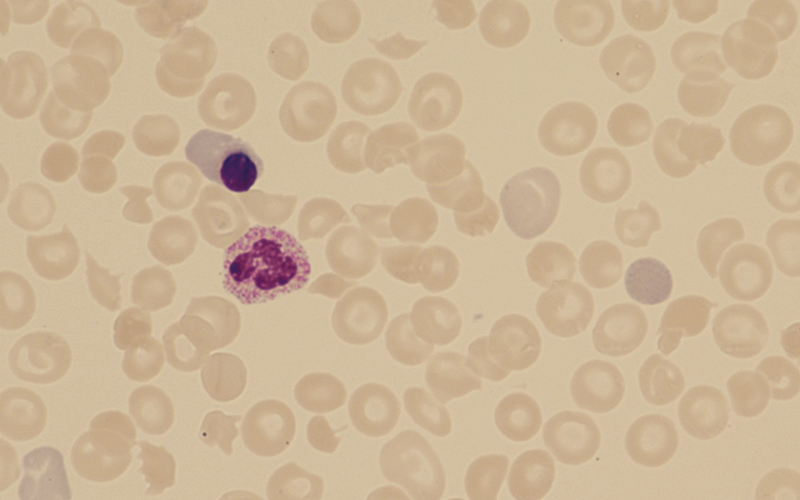
Figure 5 (patient 3): ×600 magnification. Day 14 shows significant fall in haemoglobin with scanty fragments seen. C. canimorsus remain within some of the neutrophils (arrowed).
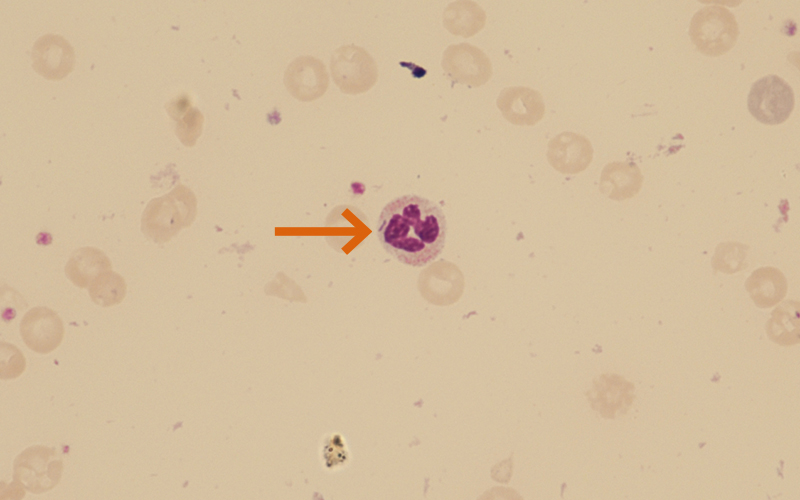
Patient 3 Day 9
- Hb 77 g/L (130–180)
- WBC 19.2 x109/L (4.0–11.0)
- Platelets 13 x109/L (150–450)
- Neutrophils 15.1 x109/L (2.0–7.5)
- D-dimer 19,003 ng/mL FEU (1–499)
- Fibrinogen 0.61 g/L (1.9–4.8)
- CRP 272 mg/L (0–5)
- Urea 31.7 mmol/L (2.5–7.8)
- Creatinine 334 µmol/L (59–104)
Patient 1
Patient 1, a 67-year-old male with a history of dog bites, presented two weeks earlier. The blood film confirmed marked thrombocytopenia, and neutrophils showed marked vacuolation and demonstrated scanty intracellular rod-shaped structures. These were also seen extracellularly. The patient was immunocompetent, received antibiotics for localised cellulitis, and was discharged within eight days with no lasting consequences from the infection.
Patient 2
Patient 2, a 49-year-old male, presented to his GP feeling unwell one week following “penetrating dog bites”. FBC, U&E and CRP were requested with FBC showing marked normocytic normochromic anaemia, mild leucocytosis, and mild thrombocytopenia. The blood film showed significant red cell abnormalities including schistocytes. Kidney function was normal, with the CRP markedly raised. The GP was contacted, and the patient was admitted urgently as a microangiopathic haemolytic anaemia was suspected on a background of severe infection. Patient 2 deteriorated rapidly and was admitted to ITU within 24 hours with sepsis and confirmed DIC. Daily blood films continued to show C. canimorsus and scanty blue-green inclusions within the cytoplasm of the neutrophils, in addition to the presence of Dohle bodies.
Blue-green neutrophilic inclusion bodies are a rare morphological finding closely associated with increased risk of morbidity and mortality. They are seen in patients with organ failure, more specifically acute hepatic failure, lactic acidosis, and septic shock. In a series of twenty patients reviewed, 65% of the patients died within days of the detection of the inclusions. They are most frequently associated with infection by Escherichia coli but can be seen in sepsis and organ failure caused by other organisms such as C. canimorsus. It is thought that the blue-green inclusions likely originate from lipofuscin – yellow-pigmented granules of lipid-containing residues of lysosomal digestion. Lipofuscin can be found in a number of organs and structures including the liver, heart and kidneys. In current hypotheses, acute hepatic failure secondary to hypoxic injury results in the release of lipofuscin from necrotic hepatic parenchymal cells with macrophages and Kupffer cells then taking up the pigmented granules, resulting in the formation of blue-green inclusion bodies. Following a prolonged ITU stay, patient 2 was stepped down and eventually discharged.
Patient 3

Patient 3, a 52-year-old male, presented to A&E with a history of feeling unwell following a dog bite one week prior. The presentation FBC showed normal red cell indices, mild leucopenia that initially persisted, and genuine marked thrombocytopenia (24 x109/L). Blood film showed reactive changes in the neutrophils consistent with a bacterial infection, i.e. toxic granulation and vacuolation. At this point no neutrophilic inclusions could be seen, and there were no schistocytes on the blood film. Routine coagulation screening on admission showed a mildly elevated INR, with raised D-dimer (8,748 ng/mL FEU) and low fibrinogen (0.94 g/L).
The patient deteriorated rapidly over the next few days with daily blood films being reviewed for the presence of schistocytes. The blood film on day 3 (Figure 4) showed a leucoerythroblastic picture with neutrophils with toxic granulation, occasional nucleated red blood cells, and significant red cell changes could now be seen.
Patient 3 developed a significant coagulopathy with a markedly raised D-dimer that persisted for the length of the ITU stay. IIt is recognised that sepsis can cause a coagulopathy to occur, taking the form of sepsis-induced coagulopathy (SIC) or sepsis-associated DIC, as seen in patients 2 and 3. The British guidelines for the diagnosis and management of DIC state that “the cornerstone of the treatment of DIC is treatment of the underlying condition” with early antibiotic therapy key for sepsis patients. Despite significant intervention patient 3 continued to deteriorate, developed acute renal failure, gangrenous digits, and sadly passed away after six weeks.
It has been suggested that C. canimorsus uses several mechanisms to evade the immune system, explaining its lethal pathogenesis. Studies have shown that C. canimorsus does not activate signals that lead to the release of pro-inflammatory cytokines, chemokines, and nitric oxide. In one study, human macrophages were unable to mount a potent inflammatory response when infected with C. canimorsus. It has also been reported that the lipopolysaccharides of C. canimorsus protect against deposition of complement membrane attack complex and prevent phagocytosis by neutrophils. It is clear from the images above though, that phagocytosis by neutrophils can occur.
Diagnosis of C. canimorsus is usually made using blood cultures, however, it is recognised that identification using this method can be problematic – in a US study only one third of Capnocytophaga isolates were correctly identified by submitting laboratories. It is likely this is due to the slow-growing nature of C. canimorsus and other methods such as PCR, 16S rRNA gene amplification, and mass spectrometry should be utilised instead.
Conclusion
It is essential that in patients with bacterial sepsis, who have a history of cat or dog bites or exposure to these animals, C. canimorsus is suspected as a potential source of infection as early as possible so that the high morbidity and mortality rates associated with this infection can hopefully be prevented.
Nicki Lawrence is Principal Biomedical Scientist Advanced Practitioner in Morphology at North Midlands and Cheshire Pathology Service. All cases featured have been fully anonymised. To view the article with full references, visit thebiomedicalscientist.net
Image credit | iStock




