This series continues with a brief review of pioneering work in the development, analysis and clinical application of important tumour markers, with two examples from the CA serum series – CA125 and CA15-3.
In a previous article in this series, tumour markers were defined as substances released by a tumour into blood or urine, or from the host in response to the tumour. Their measurement in serum or urine may be used in screening asymptomatic patients for diagnosis, prognosis, monitoring treatment and recurrence detection.
CA125 – an ovarian cancer marker and analysis
The existence of ovarian tumour-associated antigens was established by a US team led by Michael Levi in 1969 using Ouchterlony double diffusion and gel filtration (1).
In 1974, Suzanne Knauf and her colleagues used immunodiffusion and immunoelectrophoresis to demonstrate antigens in three subtypes of ovarian cancer. Four years later, Bhattacharya and Barlow detected an ovarian cystadenocarcinoma-associated antigen specific to serous or mucinous types of ovarian cancer, which was measured by radioimmunoassay inhibition and found to be raised in the sera of ovarian cancer patients (2). In a landmark paper in 1981, with Robert Bast Jr leading a US combined research team at Brigham Hospital and Harvard, Boston reported the development of a highly specific murine monoclonal antibody against human ovarian cancer. The 125th most promising clone was termed OC125 and the reacting antigen CA125 (3). A serum radioimmunoassay using OC125 was developed by Bast and colleagues in 1983 to monitor epithelial ovarian cancer.
From 1991, immunometric assays became available, often using two epitopes, one recognised by OC125 and the other by M11, an antibody developed by Tim O’Brien and colleagues at Arkansas (4). This assay, designated CA125II, showed improved assay precision. Commercial assays from seven major suppliers were compared in 1998 and found to be clinically reliable for quantitation of CA125 (5). Modern automated two site immunoassays use-high sensitivity fluorimetric and chemiluminescent tracer detection assays. It has been proposed that the major forms of CA125 in serum have molecular weights 200- 400kDa and the most often quoted serum reference range for females is 0-35kU/L (0-35 IU/ml). However, it is significant that this value is exceeded in many other malignancies, such as pancreas, liver and lung and several “benign” conditions, e.g. uterine fibroids and endometriosis.
CA125 structure and function
CA125 is a high molecular weight glycoprotein epitope on a large transmembrane mucin glycoprotein MUC16, but due to its complexity and mucinous nature, there are conflicting views on its true molecular structure (6). The MUC16 gene is expressed under normal conditions in epithelial tissues, such as breast and lung, but overexpressed in epithelial cancers. It has been proposed that it may have a lubricating function and protect against foreign particles and infectious agents. It may also act in a more sinister manner to promote cancer cell proliferation and metastases, and inhibit anti-cancer immune responses (7).
CA125 and ovarian cancer
Ovarian cancer is the sixth most common UK female cancer, with a low prevalence, particularly in premenopausal women. Often, vague symptoms lead to a late diagnosis and a relatively poor prognosis, with a 46% five-year survival rate (8). Risk factors include age, a close family history of ovarian cancer and inherited mutations in the BRCA1 and BRCA2 genes.
CA125 is only raised in 50% of stage 1 patients which, with low specificity, severely limits its role in screening (9). Nevertheless, there have been a number of clinical trials in postmenopausal women involving serum CA125 alone, or combined with transvaginal ultrasound or a variety of risk algorithms. The largest randomised controlled trial of over 200,000 postmenopausal women in UK took place in 2001, led by Ian Jacobs and Usha Menon at University College London with three study groups – CA125 and risk algorithm, annual transvaginal ultrasound, or no screening. It was claimed that the results suggested that there may be a reduction in mortality with CA125 and risk algorithm (10).
However, the main clinical roles of serum CA125 are twofold: to differentiate benign and malignant pelvic masses in postmenopausal women combined with ultrasound and, secondly and most notably, serial measurements to monitor the response to chemotherapy treatment following surgical resection and the subsequent detection of recurrence (11). Other proposed biomarkers for ovarian cancer include HE4 – human epididymis protein 4, which may be combined with CA125 to improve both sensitivity and specificity, and mesothelin – a small protein which is over expressed in ovarian cancer (12).
Breast cancer
Breast cancer has been known from ancient times but most progress was made during the 19th and 20th centuries, notably following the US National Cancer Act in 1971 with its generous funding for cancer research (see table for the most significant achievements).
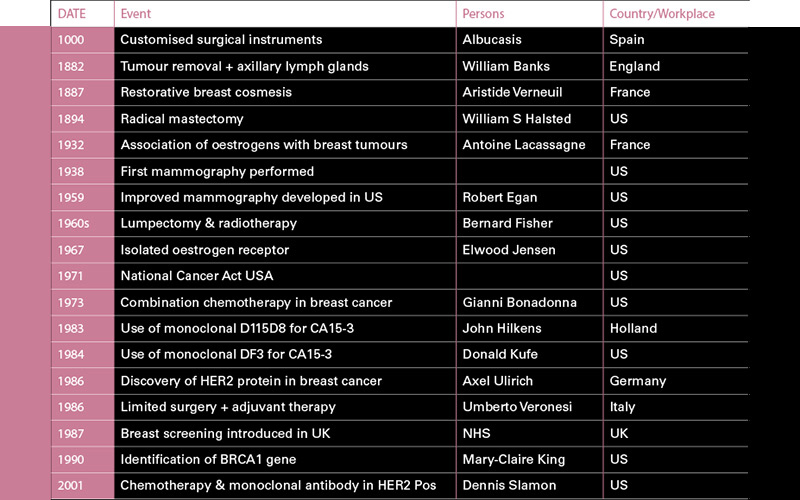
Despite all this valuable research, it remains the most common cancer in women worldwide and the second most common cause of cancer death in females in the UK. However, it is more encouraging that with early and personalised treatment, 87% of breast cancer patients have a five-year survival rate. Tumours may occur in the breast ducts or less often affect lobular cells, and around 80% of invasive breast cancers are ductal and metastases may result in the lungs, bones or brain.
CA15-3 a serum tumour marker for breast cancer
Studies, notably in the US and Italy by ME Edynak (1971) and G Fossati (1972) respectively, helped to establish the existence of tumour-associated antigens in breast cancer (13). It was further shown that the antigen could be characterised by sequential solubilisation, protein fractionation and polyacrylamide agar gel electrophoresis. Much was gained for our understanding of the structure of the glycoprotein MUC-1 associated with breast cancer by the studies of the British biologist and geneticist Joyce Taylor-Papadimetriou. MUC-1 is a large transmembrane mucin glycoprotein encoded by the MUC-1 gene and is present in normal secretory epithelia acting as a preventive barrier to pathogenic invasion but may also play an active role in oncogenesis and is overexpressed in breast cancer and shed into the circulation. One antigen on MUC-1, known as CA15-3, can be measured using a variety of immunological methods using the murine monoclonal antibodies, 115D8 & DF3, developed by J Hilkens and colleagues (1983) and a group led by D Kufe (1984), respectively (14,15). With further developments during the next decade, automated two site immunoassays became available using a variety of capture and detection systems for serum CA15-3. With assays, more robust and sensitive numerous clinical trials were set up to assess clinical values. A number of tumour markers for breast cancer, including CA15-3, were reviewed in 1993 and found that CA15-3 reflected tumour burden and was clinically useful in monitoring therapy (16). On a technical note, the standard reference range for females is <30 U/ml, and some patients exposed to mouse antigens within the environment, treatment or imaging procedures may have mouse antibodies which interfere in the assay of CA15-3.
Clinical trials of serum CA15-3
A vast number of clinical trials of variable size and quality have been performed during the last 30 years relevant to tumour markers in breast cancer and it is only possible in a short review to present a general consensus of results; in addition, various expert oncology groups in Europe and the US have made recommendations with regular updates.
The early trials of the late 80s and early 90s were optimistic of the clinical value of CA15-3; however, it was quickly recognised that it was another nonspecific marker with low sensitivity in preoperative breast cancer and unsuitable for screening asymptomatic patients. Elevated serum levels were found in a number of non-mammary cancers, such as lung, colon and ovary, and in benign breast and ovarian conditions. Sensitivity was found to be 10-15%, 20-25% and 30-35% for stages 1, 2 and 3, respectively (17). It had been previously shown that increasing levels may be useful in detecting advanced disease, and combination with serum carcino-embryonic antigen (CEA) allows early diagnosis of metastases in 60-80% of patients with advanced disease (18).
Expert group the American Society of Clinical Oncology released a 2007 update that reports present data is insufficient to recommend serum CA15-3 for screening, diagnosis and staging and for monitoring recurrence after primary breast cancer therapy, but permits monitoring of metastatic disease during active therapy when combined with diagnostic imaging, history and physical examination (19). Later reports and recommendations tend to limit CA15-3 to this clinical application and propose the use of oestrogen receptor tissue assays to predict the response to hormone treatment and the measurement of human epidermal growth factor receptor 2 (HER2) to identify patients to respond to combined monoclonal antibody therapy, such as that with Herceptin (8).
Concluding comments
While much has been achieved in the diagnosis and treatment of ovarian and breast cancer, the role of both tumour markers reviewed appears limited to monitoring treatment. The focus of attention in breast cancer patients at present is on the results of the (TAILORx) trial, using a combination of HER2 results and a 21-tumour gene expression microarray to determine the benefits or otherwise of adjuvant chemotherapy.
The research performed by Robert Bast, Joyce Taylor Papadimitriou, Mary-Claire King, Donald Kufe and John Hilkens was fundamental in achieving significant progress during the last four decades.
Stephen Clarke is a retired IBMS Fellow.
Image Credit | Science Photo Libary
References
1. Levi MM, Keller S, Mandl I. Antigenicity of a papillary cystadenoma tissue homogenate and its fractions. Am J Obs Gynae 1969; 105(6): 856-861
2. Bhattacharya M, Barlow JJ. Ovarian Tumour antigens. Cancer 1978; 42 :1616-1620
3. Bast R Jr, Feeney M, Lazarus H et al. Reactivity of a monoclonal antibody with human ovarian cancer. J Clin Invest 1981; 68: 1331-1337
4. Diamandis E, Bast R Jr, Gold P et al. Reflections on the discovery of carcino-embryonic antigen, prostate specific antigen & Cancer antigens CA125, CA19-9. Clin Chem 2013; 59(1): 22-31
5. Davelaar E, Van Kamp GJ et al. Comparison of seven immunoassays for the quantification of CA 125 antigen in serum. Clin Chem 1998; 44(7): 1417-1422
6. Felder M, Kapur A, Gonzalez –Bosquet J et al. MUC16 (CA125): Tumour marker to cancer therapy, a work in progress. Mol Cancer 2014; 13: 129-
7. Rump A, Morikawa Y, Tanaka M et al. Binding of ovarian cancer antigen CA125/MUC16 to mesothelin mediates cell adhesion. J Biol Chem 2004; 279: 9190-9198
8. Garner EI, Garrett A et al. Natural History and detection of ovarian cancer. Glob.libr women’s med 2008. GLOWM 10246
9. Duffy MJ Tumour markers in clinical practice: A review focusing on common solid Tumours. Med Princ Pract 2013; 22:4-11
10. Jacobs I, Menon U et al. Ovarian cancer screening and mortality in the UK Collaborative trial of ovarian cancer screening: a randomised controlled trial. Lancet 2016; 387(10022): 945-956
11. Duffy MJ, Bonfrer JM et al. CA125 in ovarian cancer: European Group on tumour markers guidelines for clinical use Int J Gynaecol Cancer 2005; 15(5): 679-691
12. Yang WL, Lu Z, Bast R Jr. The role of biomarkers in epithelial ovarian cancer. Expert Rev Mol Diagn 2017; 17(6): 577-591.
13. Neville AM Clinical value of Tumour associated antigens J Clin Path 1974; S3-7: 119-126
14. Hilkens JL, Buijs F, Hilgers J. Monoclonal antibodies against human milk-fat globule membranes detecting differentiation antigens of the mammary gland and its Tumours. Int J Cancer 1984; 34(2): 197-206
15. Kufe D, Inghirami G, Abe M et al. Differential reactivity of a novel monoclonal antibody (DF3) with human malignant versus benign breast Tumours. Hybridoma 1984; 3(3): 223-232
16. Werner M, Fazer C, Silverberg M. Clinical utility and validation of emerging biochemical markers for mammary adenocarcinoma Clin Chem 1993 ; 39(11) : 2386-2396
17. Duffy MJ. Serum Tumour markers in breast cancer: are they of clinical value. Clin Chem 2006; 52(3): 345-351
18. Mirabelli P, Incoronato M. Usefulness of traditional serum biomarkers for the management of breast cancer patients. Biomed Research International 2013, Article ID 685641
19. Harris L, Fritsche H et al. ASCO 2007 Update in recommendations for the use of Tumour markers in breast cancer. J Clin Oncol 2007; 25(33): 5287-5312.
Table references
1. Lakhtakia R. A brief history of breast cancer. Sultan Quaboos Uni Med. 2014; 14(2): 166-169
2. Ades F, Tryfonidis K, Zardavas D, The past and the present breast cancer treatment, Ecancermedicalscience 2017; 11 746




