Following the completion of a high-quality audit, the team at Manchester Royal Infirmary’s cytology department offer guidance on monitoring a clinical service.
With the recent implementation of ISO standards to laboratory accreditation, audits can be a useful tool in examining the clinical service streams and service monitoring. It is a method of continuous quality improvement that allows for systematic reflection and review of practice. It is important that audits:
- Are measured against relevant, established standards
- Provide feedback to service users/groups involved in the practice
- Promote intervention/training, if required
- Are followed up to see if recommendations are in place and working.
The non-gynaecological (NG) cytology department of Manchester Royal Infirmary has been providing a biomedical scientist (BMS) on-site specimen preparation and adequacy assessment for fine needle aspirations (FNAs) taken at all head and neck (H&N) clinics for a number of years. Best practice implementation, training and audit of this service has resulted in specimen adequacy rates of over 90%.
In 2013, a new work stream came online when a local district hospital merged with the trust. The H&N service at this hospital did not have BMS on-site assistance, or adequacy assessment and, as it was not cost-effective to implement such a service at the time, there were initial discussions regarding sample collection and transport and reporting.
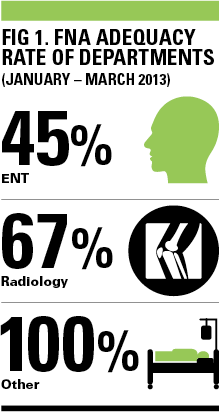
This was because the neck lump clinics were incorporated into daily ear, nose and throat (ENT) clinics. This H&N service was then audited within three months with disappointing results of an adequacy rate of 45% (see Fig 1). An action plan was created with the recommendations:
- Feedback to clinical director and teams.
- Practical training sessions for clinicians by the NG cytology team to promote best practice on how to obtain FNA samples and prepare spreads.
- FNAs should be performed under ultrasound guidance if possible, as it was observed that samples taken under radiological guidance by the radiologists had an adequacy rate of 67%.
- Plan to re-audit in one year’s time to check for improvement.
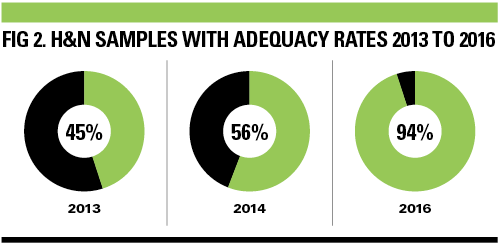
However, the re-audit (January to March 2014) showed only a marginal increase in the adequacy rate of H&N samples (56%).
The ENT department then employed a specialist ENT surgeon who requested a dedicated neck lump clinic for one afternoon per week. With this in mind, the cytopathology department was able to provide a BMS for on-site assistance and adequacy assessment for the H&N samples and it was agreed that the surgeon would aspirate the samples using radiological guidance. The BMS on-site assistance included:
- Receiving the needle safely from the surgeon and preparing two direct spreads per aspiration (x1 air dried, x1 alcohol fixed).
- Rinsing remaining needle material in a fixative.
- Staining one/both air dried spread and performing adequacy assessment. This information is then immediately conveyed to the surgeon who would then perform another one to two aspirates for samples deemed inadequate or non-diagnostic.
The service started in December 2015 and the plan was to audit January to March 2016.
The re-audit was approached slightly differently, as it was decided that published data should be used to show how the service was performing and whether full compliance could be given in line with the trusts audit report criteria. It was difficult to find an established standard for H&N FNA adequacy rates.
The Royal College of Pathologists suggests achievable FNA thyroid adequacy rates should be between 69% and 97% with training, but no noted adequacy rates seem to be available for other H&N FNA cytology samples. A literature search found a meta-analysis from the Royal College of Radiologists that suggested the average adequacy rate for all H&N FNA cytology samples is 90.7%. This was used as the standard for the re-audit of 2016. The results of 2016 re-audit of the H&N samples following these changes showed there had been an improvement in adequacy rates to 94% (see Fig 2).
The findings of the first and second audits suggested many of the H&N specimens were taken without radiological guidance and by various clinicians throughout the week.
However, with the implementation of a dedicated lump clinic, with samples taken by an experienced surgeon under radiological guidance, together with on-site BMS assistance and specimen adequacy assessment, there was a marked improvement.
It will be important to re-audit in the future to see if these high standards are maintained and to check that the improvements have had the desired and expected effect.
LESSONS LEARNT
- Even though an improvement has been made, it is important to remember that standards need to be maintained and doing audits in this way can highlight problems and help with solutions.
- Communication with the ENT and cytology departments helped to achieve an organised weekly clinic with excellent team work and improved service and adequacy rates with direct patient benefits.
- It is important to always question whether a service identifies and promotes good practice and whether it leads to improvements
- in patient care.
- Good practice can be shared locally and nationally.
David Slater and colleagues, Cytology Centre, Manchester Royal Infirmary




