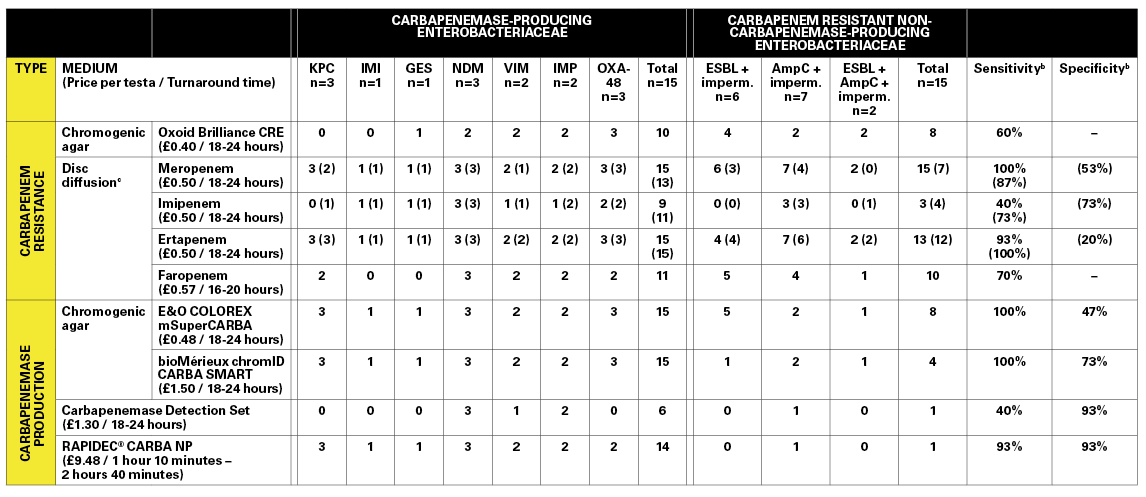
A comparison of phenotypic methods for the screening of carbapenem-resistant and carbapenemase-producing Enterobacteriaceae.
Antibiotic resistance is now frequently seen in Enterobacteriaceae and the therapeutic choices for treating multidrug-resistant bacteria can be limited. As such, a shift in the empirical choice of antibiotics has led to an increasing reliance on carbapenems and subsequently, a rise in carbapenem-resistant Enterobacteriaceae (CRE). CRE can occur through the acquisition of a carbapenemase, and patients with such carbapenemase-producing Enterobacteriaceae (CPE) have a significantly increased mortality over those who do not: 59.5% vs. 27.4%, and 78% vs. 11%. This increase, and the potential for CPE to disseminate rapidly in hospitals, underpins the importance of screening for CPE.
This study evaluated primary screening methods for carbapenem resistance, and, where applicable, carbapenemase production, while considering efficacy, cost, turn-around time, expertise required and the ease of its potential implementation.
Thirty non-duplicate carbapenem-resistant Enterobacteriaceae strains were obtained from Public Health England. Fifteen strains harboured a carbapenemase and the other 15 strains were identified as carbapenem-resistant through other mechanisms.
Strains were screened blind, alongside EUCAST recommended control strains using three chromogenic agars: one designed for detecting carbapenem resistance (Brilliance™ CRE, Thermo Fisher Oxoid Ltd), and two designed for detecting carbapenemase production (COLOREX™ mSuperCARBA, E&O Laboratories Ltd; and chromID® CARBA SMART, bioMérieux). Each agar was tested in triplicate with a 1μl aliquot inoculum of a 0.5 (+/-0.05) MacFarland standard suspension made in saline, and incubated aerobically at 37°C for an initial 18 hours before re-incubating for a further six hours in absence of growth. Carbapenem resistance was also screened for via disc diffusion using meropenem, imipenem and ertapenem (10µg disc content), and CAT-ID faropenem discs (D71C, Mast Group Ltd). Inhibition zones for meropenem, imipenem and ertapenem were measured and interpreted according to BSAC carbapenemase screening guidelines. Additionally, carbapenemase production was screened for using Carbapenemase Detection Set discs (D70C, Mast Diagnostics), whereby zones of inhibition were measured and interpreted according to manufacturer’s instructions. Carbapenemase production was also screened for using the RAPIDEC® CARBA NP test (bioMérieux), conducted according to manufacturer’s instructions.
When comparing methods designed for detecting carbapenem resistance, the results demonstrated that meropenem discs are the most reliable indicator (100% sensitivity) compared to other carbapenem discs (ertapenem, 93%; faropenem, 70%; imipenem, 40%). Amongst the carbapenemase detection methods, the COLOREX™ mSuperCARBA and the chromID® CARBA SMART were equally sensitive across all strains (100%), but struggled to be highly specific. Overall, the Carbapenemase Detection Set discs performed poorly (40% sensitivity, 93% specificity). However, the discs only claim to be able to detect KPC and metallo-β-lactamase producing strains, serving as a huge limitation of the method in the present study, and completely ruling out the method as a standalone confirmatory test for carbapenemase activity. The RAPIDEC® CARBA NP assay performed well, with one false positive (AmpC + impermeability), and one false negative (OXA-48), and therefore represented the test with the best overall performance.
For carbapenem resistance detection, the price of the carbapenem discs and Brilliance™ CRE agar was similar, with similar lengths of time to produce a result. For the carbapenemase detection methods, the RAPIDEC® CARBA NP was the most expensive; however, it was straightforward to conduct and yielded results in a maximum of two hours and 40 minutes, compared to 18 to 24 hours for the other methods. We believe this justifies its higher cost per test, as a reduced turn-around time could assist in the rapid de-escalation of ad ditional infection control measures where suspected cases of CPE are proved negative. Furthermore, the very mechanism of the test is fundamental to its success in that it detects carbapenem hydrolysis and is not limited to a smaller subset of carbapenemases defined by the manufacturer.
Since this study was conducted, the BSAC susceptibility testing guidelines that were used have been replaced with EUCAST guidelines. If the EUCAST CPE screening guidelines are applied to the disc diffusion data from this study, meropenem discs still provide the best balance between sensitivity and specificity (see Table, results in brackets). However, the difference in recommended susceptibility testing agar between the two sets of guidelines limits the ability to apply EUCAST cut-offs to these data.
(Click to enlarge)
To conclude, we used a two-step algorithm that could be employed to screen patients for CRE, and then any positive samples for CPE. We found meropenem to be the best indicator of CRE, while the RAPIDEC® CARBA NP test gave the best balance between sensitivity and specificity in the confirmation of CPE. While more expensive, the RAPIDEC® CARBA NP test allows for potential savings to be made in the wider hospital through a rapid turnaround time. Both the meropenem discs and the RAPIDEC® CARBA NP test would adapt well to a generalised screening algorithm in our hospital in the future, which may move towards screening for carbapenem resistance in other organisms, such as Pseudomonas spp. and Acinetobacter spp.
Cherie D Beckett (Cherie.beckett@ pah.nhs.uk), Biomedical Scientist at Princess Alexandra Hospital NHS Trust, with Katie L Hopkins, Danièle Meunier, Deborah Orriss and Benjamin A Evans.
Picture credit: CDC