Malcolm Needs CSci FIBMS, formerly of the NHSBT, delves into the history and science of a notable blood group system.

Lea was first described in 1946 by Mourant1, when it was named L. Leb was first described in 1948 by Andresen2.
In 1955, Sneath and Sneath3 observed that red cells lacking Lea and Leb will take up these antigens from plasma containing them. Equally, red cells expressing either Lea or Leb will give these up to plasma lacking them. In other words, the antigens were found to be soluble.
Between 1948 and 1951, Grubb4,5 and Brendemoen6 independently observed that the saliva of Le(a+b-) individuals strongly inhibited anti-Lea, and that the saliva of the majority of Le(a-b+) individuals also inhibited anti-Lea, but did so less strongly.
In 1963, Mollison et al7 demonstrated that this phenomenon also occurred in vivo.
From this it can be seen that:
- Lewis antigens are not intrinsic to red cells.
- They are located on type 1 glycosphingolipids that are adsorbed onto the red cells from the plasma.
- Lewis, therefore, is not strictly speaking, a red cell blood group!
Genotypes and phenotypes and their relationship with the Secretor gene
At a basic level, if you do not inherit a Lewis gene (LE, or, as it is now named, FUT3), whether you inherit a Secretor gene (SE, or, as it is now named, FUT2) of not, you will be Le(a-b-). if you do inherit a Lewis gene, but you do not inherit a Secretor gene, you will be Le(a+b-). If you inherit a Lewis gene, and you inherit a Secretor gene, you will be Le(a-b+) (see Table 1).
That having been said, the Secretor gene actually defines whether an individual secretes A, B and/or H Substance (Type 1 A, B and/or H substance) in saliva and other body fluids, rather than directly to do with the Lewis types.
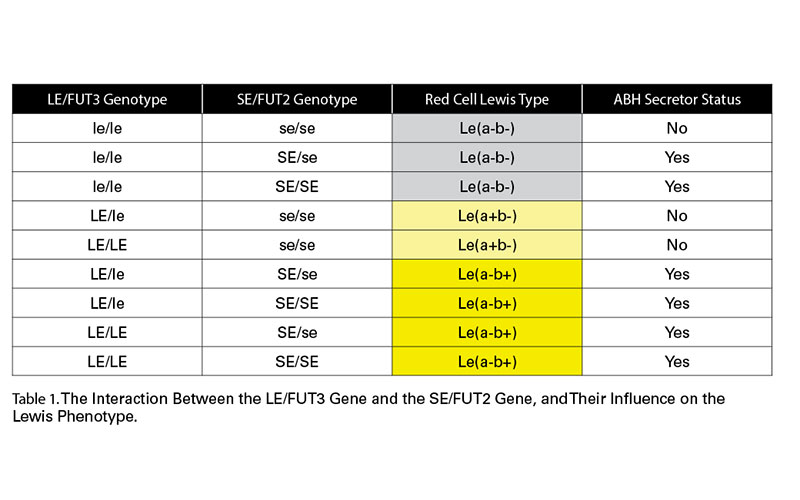
It will be noted that there is no Le(a+b+) shown in Table 1, and the reason for this will be explained below.
The genes
The locus for the gene coding for LE/FUT3, has been mapped to 19p13.38.
It was soon realised that there was an interaction between the LE/FUT3 gene and the SE/FUT2 gene (see Table 1).
The locus for the gene coding for SE/FUT2 is also found at 19p13.38, but, although they are mapped to the same chromosome, LE/FUT3 and SE/FUT2 segregate independently9-11.
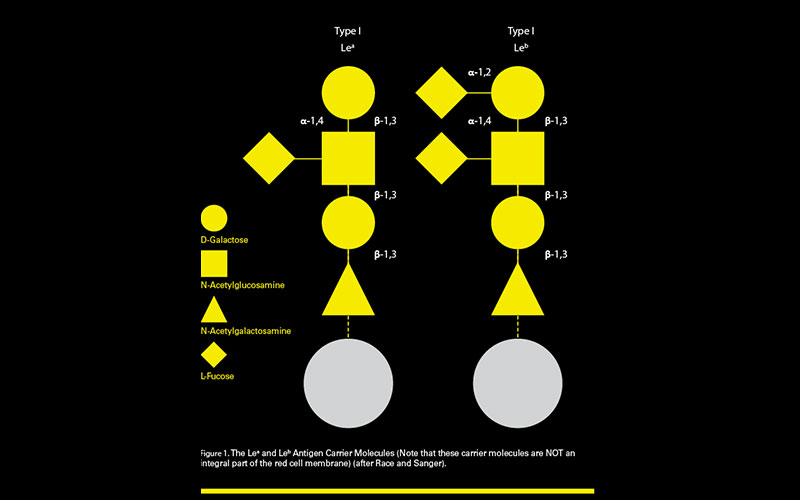
The Lewis carrier molecule
As stated earlier, the Lewis antigens are not an integral part of the red cell membrane, but are plasma soluble molecules. They are also carbohydrate-based molecules, and so, like the A, B and H antigens, they are not direct gene products. The gene products are α-1-4-fucosyltransferase (LE/FUT3) and α-1-2-fucosyltransferase (SE/FUT2). The SE/FUT2 direct gene product cannot function, unless the LE/FUT3 direct gene product is present (rather in the same way that the A and B gene products cannot function, unless the H gene product is present and functioning.
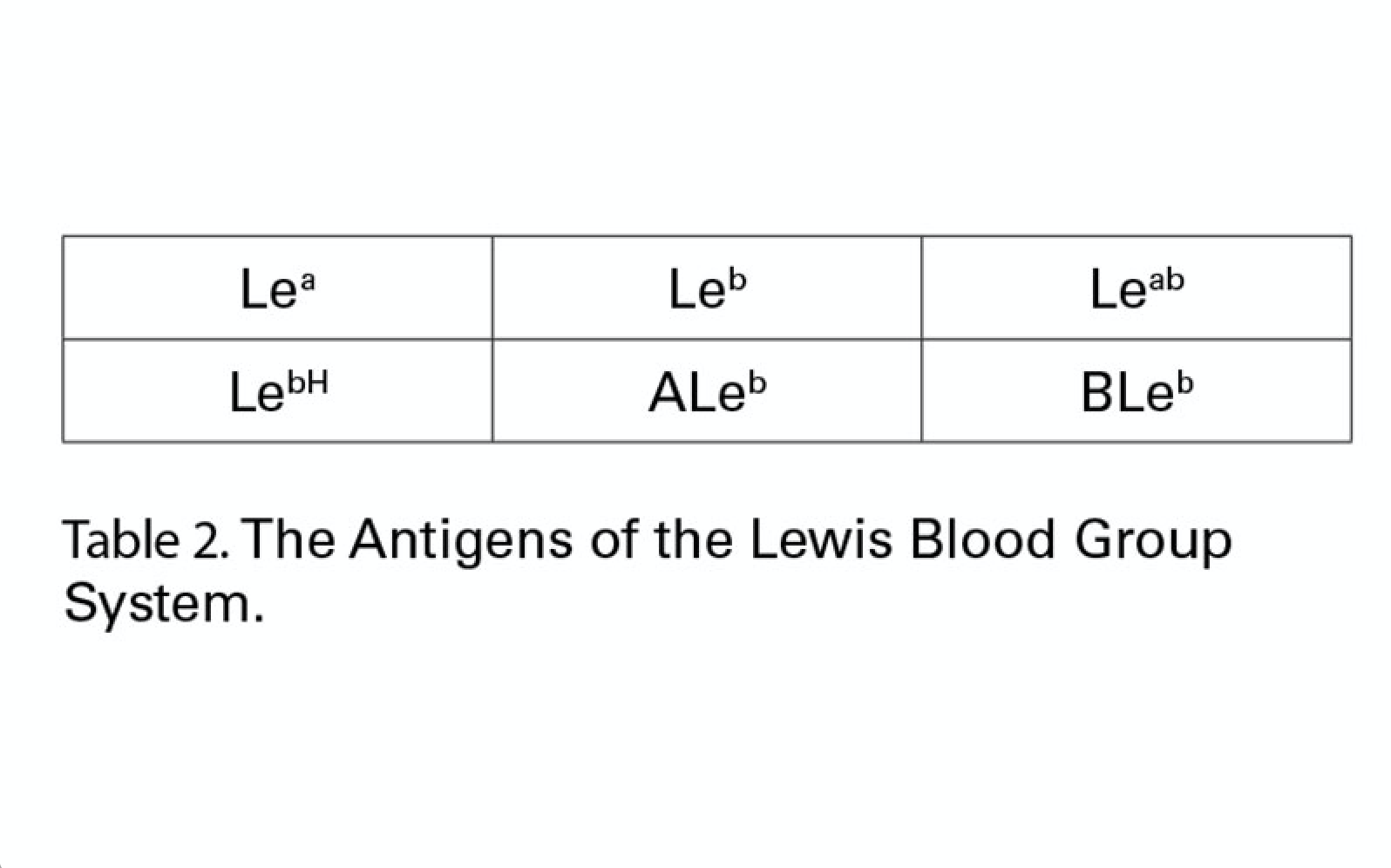
There are six antigens recognised by the International Society of Blood Transfusion (ISBT) within the Lewis Blood Group System. These can be seen in Table 2.
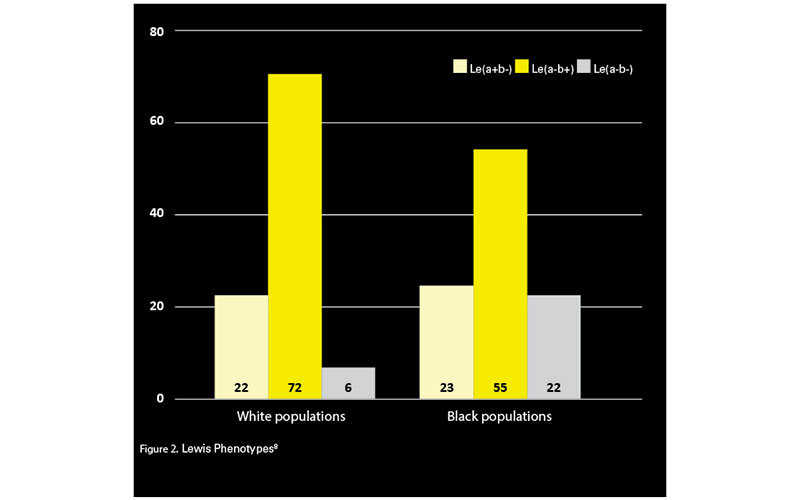
Lewis phenotype frequencies
The normal figures for Lewis types can be seen in Figure 2; however, for various reasons, such as infancy and pregnancy (see below for explanations), such figures should only be taken as true for individuals from the age of approximately two and upwards, and who are not pregnant.
Lewis antigens in newborns and infants13
Most newborn babies type as Le(a-b-) for the first month of their life, as the production of Lewis fucosyltransferase is at very low levels. If they are going on to eventually become Le(a-b+), they will, for a time, type as Le(a+b-), as the production of Lewis fucosyltransferase becomes active before Secretor fucosyltransferase. For a very short time, they may type as Le(a+b+), before they become Le(a-b+). By one year of age, 50% of children express their adult phenotype14, and by two years of age, the Lewis phenotype of most children will reflect their SE/FUT2 and LE/FUT3 alleles15.
Lewis antigens in pregnancy13
The Le(a+b+) phenotype in adults
In truth, in almost every case of the phenotype Le(a-b+), a small amount of Lea antigen can, in fact, be detected17. The frank Le(a+b+) phenotype in Polynesian adults is, however, quite common (~40%). This is due to a weak or mutated Secretor status18.
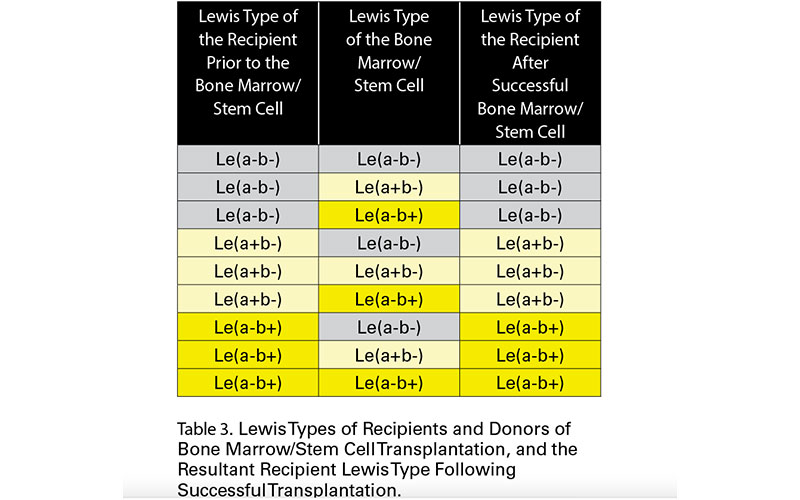
Lewis phenotypes following transplantation
As has been mentioned above, Lewis antigens are not produced as an integral part of the red cell membrane, but are adsorbed onto the red cell membrane. In 1986 and 1987, Myser et al19 and Needs et al20 independently observed that the Lewis antigens always remain of the recipient type after bone marrow transplantation.
Following successful bone marrow/stem cell transplantation all red cell antigens that are integral to the red cell membrane (i.e. are produced in the bone marrow) change from that of the recipient, to that of the donor, although, of course, these are sometimes identical. However, those that are not integral to the red cell membrane (i.e. are produced remote from the bone marrow) remain as the antigen type of the recipient. These include the soluble A, B and H antigens21, the Lewis antigens and the antigens within the Chido/Rodgers Blood Group System.
In the case of the Lewis antigens, the final Lewis phenotypes can be seen in Table 3.
 Lewis antibodies
Lewis antibodies

IgM anti-Lea is more frequent than IgG. IgG anti-Lea can cause a haemolytic transfusion reaction, albeit very rarely and self-limiting, as the Lea antigen elutes from the transfused red cells and the anti-Lea is then inhibited by this eluted antigen.
Anti-Lea has only been reported once as causing mild HDFN22.
IgM anti-Leb is more frequent than IgG. IgM and IgG anti-Leb were thought to be clinically benign, but there has been a recent report of an acute haemolytic transfusion reaction due to anti-Leb23.
There has been one (dubious) report of mild HDN caused by anti-Leb24.
There is some evidence that Lewis antibodies may be involved in renal transplant rejection, but the evidence is slight and the theory controversial25.
Malcolm Needs was the Reference Service Manager at NHSBT-Tooting Centre and is an IBMS Advisory Panel member.
To see the references, view the article online at thebiomedicalscientist.net
Image credit | Science Photo Library
References.
- Mourant AE. A ‘new’ human blood group antigen of frequent occurrence. Nature 1946; 158: 237-238.
- Andresen PH. The blood group system L: a new blood group L2. A case of epistasy within the blood groups. Acta Path Microbiol Scand 1948; 25: 728-731.
- Sneath JS, Sneath PHA. Transformation of the Lewis groups of human red cells. Nature 1955; 176: 172.
- Grubb R. Correlation between Lewis blood groups and secretor status in man. Nature, Lond 1948; 162: 933.
- Grubb R. Observations on the human blood group system Lewis. Acta Path Microbiol Scand 1951; 28: 61-81.
- Brendemoen OJ. Studies of agglutination and inhibition in two Lewis antibodies. J Lab clin Med 1949; 34: 538-542.
- Mollison PL, Polley MJ, Crome P. Temporary suppression of Lewis blood-group antibodies to permit incompatible transfusion. Lancet 1963; 1: 909-912.
- Reid ME, Lomas-Francis C, Olsson ML. The Blood Group Antigen Factsbook. 3rd edition, 2012, Academic Press.
- Ceppellini R, Siniscalco M. Una nuova ipotesi genetica per il sistems Lewis secretore e suoi riflessi nei riguardi di alcune evidence di linkage con altri loci. Revista dell’ Istituto Sieroterapico Italiano 1955; 30: 431-445.
- Sanger R, Race RR. The Lutheran-secretor linkage in Man: support for Mohr’s findings. Heredity 1958; 12: 513-520.
- Sneath JS, Sneath PHA. Adsorption of blood-group substances from serum on to red cells. Brit med Bull 1959; 15: 154-157.
- Race RR, Sanger R. Blood Groups in Man. 6th edition, 1975, Blackwell Science Publications.
- Combs MR. Lewis blood group system review. Immunohematology 2009; 25 (3): 112-118.
- Lawler SD, Marshall R. Lewis and Secretor characters in infancy. Vox Sang 1961; 6: 541-544.
- Oriol R. ABO, Hh, Lewis and secretions: serology, genetics and tissue distribution. In: Cartron JP, Rouger P, eds. Blood Cell Biochemistry: Molecular Basis of Major Blood Group Antigens. 6th edition, 1995, New York: Plenum, pp 37-73.
- Hammar L, Mansson S, Rohr T, Chester MA, Ginsburg V, Lundblad A, Zopf D. Lewis phenotype of erythrocytes and Leb-active glycolipid in serum of pregnant women. Vox Sang 1981; 40: 27-33.
- Brendemoen OJ. Studies of agglutination and inhibition in two Lewis antibodies. J Lab clin Med 1949; 34: 538-542.
- Henry SM, Benny AG, Woodfield DG. Investigation of Lewis phenotypes in Polynesians: evidence of a weak secretor phenotype. Vox Sang 1990; 58: 61-66.
- Myser T, Steedman M, Hunt K, Strohm P, Williams M, Kennedy M. A bone marrow transplant with an acquired anti-Le(a): a case study. Hum Immunol 1986; 17: 102-106.
- Needs ME, McCarthy DM, Barratt AJ. ABH and Lewis antigen and antibody expression after bone marrow transplantation. Acta Haemat 1987; 78: 13-16.
- Hult AK, Dykes JH, Storry JR, Olsson ML. A and B antigen levels acquired by group O donor-derived erythrocytes following ABO-non-identical transfusion or minor ABO-incompatible haematopoietic stem cell transplantation. Transfusion Medicine
- Carreras Vescio LA, Torres OW, Virgilio OS, Pizzolato M. Mild hemolytic disease of the newborn due to anti-Lewisa. Vox Sang 1993; 64: 194-195.
- Irani MS, Figueroa D, Savage G. Acute hemolytic transfusion reaction due to anti-Leb. Transfusion 2015; 55: 2486-2488.
- Bharucha ZS, Joshi SR, Bhatia HM. Hemolytic disease of the newborn due to anti-Leb. Vox Sang 1981; 41: 36-39.
- Daniels G. Human Blood Groups. 3rd edition, 2013, Wiley-Blackwell.




