The IBMS has awarded five research grants this year. Here the final two successful candidates outline the work that they are undertaking.
Steven J Coles
Senior Lecturer in Biochemistry, Head of Worcester Biomedical Research Group, School of Science and the Environment, University of Worcester Development of a rapid diagnostic test for the stratification of BCAT1 overexpressing acute myeloid leukaemia
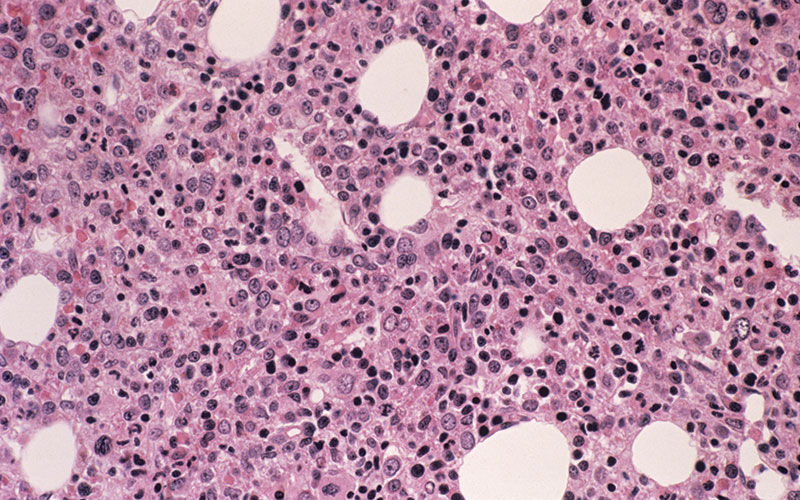
Acute myeloid leukaemia (AML) is a rapidly progressing blood cancer that manifests as a block in myeloid cell differentiation and subsequent accumulation of immature myeloid cells (blasts) in the bone marrow. Clinical signs of AML occur as a result of blast accumulation, which leads to progressive bone marrow failure. Based on genetic and clinical manifestations, AML is a heterogeneous disease according to World Health Organization classification.
Gross genetic changes, such as chromosomal translocations, can lead to the development of AML, providing these changes occur in regions of the genome that control blood cell growth and survival (e.g. t[8;21]) (RUNX1-ETO), which mutates the RUNX1 protein, a transcription factor important for
normal myeloid cell development.
Recently, the human cytosolic branched-chain amino transferase, encoded by the BCAT1 gene, has been implicated in the pathogenesis of AML. The canonical function of BCAT1 is to transfer α-amino groups from Leu, Iso and Val to α-ketoglutarate (α-KG) generating glutamate. Studies have shown that the upregulation of BCAT1 in AML cells lowers the intracellular α-KG pool, leading to epigenetic modifications. The effect mediated by BCAT1 is similar to IDH1-mutant AML. As such, BCAT1 upregulation has a negative impact on disease outcome for AML patients.
Currently there is no rapid diagnostic test for detecting BCAT1 upregulation in AML. Therefore, this IBMS Research Grant-supported project aims to develop a point-of-care diagnostic test that will allow the stratification of AML patients according to BCAT1 expression levels.
As BCAT1 may be targeted therapeutically, it is hoped that such a diagnostic test may be clinically informative in the future.
Ucheoma Ugoji
Senior Biomedical Scientist-Section Manager, Barking, Havering and Redbridge University Hospital, London Engineering and validation of catalytic nucleic acid systems for the effective diagnosis of human pathogenic Candida infections
Infections caused by fungi have become a major global problem in hospitals, with the incidence of sepsis caused by fungi increasing since the 1990s. The incidence of Candida bloodstream infection in Europe is estimated to range between 6.7 and 54 per 1,000 intensive care unit (ICU) admissions, with a mortality of more than 33.9%. In the UK, a laboratory surveillance conducted by Public Health England in 2016 showed that the overall incidence of candidaemia was 3.6 per 100,000 population. Candida infections can become disabling, cause lethal infection and have emerged as the main causes of hospital-acquired nosocomial infection, especially in immunocompromised patients and those hospitalised for long periods of time.
A range of diseases caused by fungal infections that affect healthy immunocompetent individuals generally resolve with chemotherapy, but hospital-acquired infections pose a serious threat to immunocompromised patients on hospital wards and in ICU worldwide. Current diagnosis of fungal infections relies on the isolation and identification of fungi from clinical samples, but such microbiological methods are time-consuming and lack sensitivity. Alternative fast, efficient and cost-effective diagnostic methods are needed urgently. Nucleic acids enzymes are catalytic DNA or RNA sequences that can act as biomarker sensors and molecular switches. Innovative multicomponent multiple nucleic acid enzyme (MNAzyme) systems that can identify DNA biomarkers with high sensitivity and specificity have been developed recently.
These MNAzyme systems offer new opportunities for biomarker detection and multiplexing with the potential to improve on the molecular diagnostic tools currently available. Multicomponent MNAzymes self-assemble and become catalytically active only in the presence of a biomarker target sequence and a hydrolysable fluorescent probe. If a target fungal biomarker is present in a biological sample, detection and quantification of enzymatic amplified fluorescence gives a positive diagnosis.
MNAzymes have advantages for multiplex analysis. The catalytic core and reporter arms are generic and have been designed specifically to perform optimally together without any relation to the target sequence. MNAzymes have a high conformity as they require target-specific binding of two partzymes and two PCR primers, compared to three levels of specificity associated with other multiplex assays. MNAzymes have the ability to discriminate between closely related sequence variants, and this attribute is based on the design of the target sensor arms. MNAzymes also have the ability to discriminate between single base polymorphisms, allowing detection of associated drug resistance.
The system is an improved multiplex PCR design that is specific and requires minimal optimisation.
Despite the advances made in the diagnosis and treatment of candidiasis, the infection still has high mortality rates.
The rapid detection of Candida has become very important, especially in immunocompromised patients. Current laboratory diagnostic procedures are time-consuming and require two to four days to identify Candida species.
A rapid detection and identification procedure is crucial as Candida species infect mostly preterm neonates and immunocompromised patients in ICU. The emergence of multidrug antimicrobial resistance warrants the development of a rapid detection platform for the diagnosis of Candida infection. Rapid and sensitive detection of Candida infection can play a role in the effective management of candidiasis and treatment monitoring.
This IBMS Research Grant-supported project will develop and implement MNAzymes as simple nucleic acid biosensors for the accurate, fast and reliable diagnosis of human candidiasis. With the use of isothermal DNA amplification, the research aims to develop diagnostic kits that can be used at or near the site of patient care.
Picture credit | Science Photo Library




