Ibrahim Nakibingé looks into one of the first generation of adenosine diphosphate receptor antagonists.

Plavix, commonly known as clopidogrel, is widely used as the gold standard dual antiplatelet therapy when combined with aspirin. However, the pharmacogenetic variability of the liver drug metabolising enzyme cytochrome-P450 isoenzyme CYP2C19 has been shown to be associated with reduced platelet inhibition as well as major adverse effects in a selected number of acute coronary syndrome (ACS) and myocardial infarction (MI) patients.
In the US, the Food and Drug Administration (FDA) recommends genotyping before prescribing clopidogrel to cardiovascular disease patients; however, several organisations, including the American College of Cardiology, have opposed this view, citing insufficient evidence. This paper briefly reviews the literature on CYP2C19 genotyping and responsiveness to clopidogrel to decide whether the drug is suitable for patient treatment or if there are alternatives better suited from a genomic perspective.
Background
Clopidogrel, along with ticlopidine, is considered among the first generation of adenosine diphosphate (ADP) receptor antagonists, or more conventionally referred to as P2Y12 inhibitors, as they block ADP binding to P2Y12 receptors on platelets.
The metabolism of the drug is associated with several genes including ABCB1, CES1 CYPA2, CYPB6, CYP2C9, CYP3A4, CYP3A5, P2PY12 and indeed CYP2C19. As with other CYP450 superfamily members, CYP2C19 is highly polymorphic, more than 25 known variants have been reported with subgroups being associated the enzymatic activity in clopidogrel metabolism. The technology (next generation sequencing [NGS]) to identify these variants in patients is now available to clinicians with some drug regulatory bodies sanctioning their use in tailoring patient treatment.
CYP2C19 GENOTYPING
Early CYP2C19 genotyping was based on traditional methods such as qPCR, which are heavily time-consuming and therefore unsuitable; for example, in determining the patient’s treatment course. Furthermore, due to the mutations that may occur during product amplification, the method did not yield adequate accuracy until the advent of AmpliChip CYP450 GeneChip microarrary technology which evolved from Affymetric GeneChip platform.
Based on the genotype obtained, the AmpliChip CYP450 test for CYP2C19 is able to predict the patients as poor metaboliser (PM), intermediate metabolisers (IM), extensive metaboliser (EM) or ultra-rapid metabolisers (UM). Furthermore, in a CYP2D6 study using the AmpliChip CYP(450) test, 33 variants were detected that included those associated with dysfunctional enzyme activity, while compared to traditional techniques. Depending on the NGS platform used, the application employed to identify SNPs is likely to be superior and, unlike array methods, is less likely to suffer from biased missingness that lead to false associations. Additionally, NGS technologies offer the superior targeted sequencing of exomes. Nonetheless, it is evident the genotyping technologies are sufficient enough in their ability to detect variants, and indeed the problem faced is that of interpreting the results correctly so as to allow the planning of a suitable treatment course.
Interpretation, indications and contradictions
Clopidogrel has been reported in a number of clinical studies along with other antiplatelet agents. For example, the Joint Utilisation of Medications to Block Platelets Optimally (JUMBO-TIMI(26)) and successively the Prasugrel in Comparison to Clopidogrel for Inhibition of Platelet Activation and Aggregation (PRINCIPLE) studies, both found prasugrel to significantly enhance inhibition of platelet aggregation when compared to clopidogrel. Furthermore, prasugrel’s duration of onset was faster (peak plasma concentration at 30 minutes) as it only required one CYP450 enzyme for activation, whereas clopidogrel requires two. Such differences in drug responses may or may not be associated with the gene’s polymorphic variants. A meta-analysis of CYP2C19 genotyping found that overall there is no significant association between genotype and clopidogrel responsiveness in relation to cardiovascular events. However, between certain genotypes the difference observed was significant as found between PM (CYP2C19 *2-*8) and UM (CYP2C19 *1,*17). Therefore, depending on the patient’s genotype, the dose prescribed by a clinician can vary, and it is further complicated by heterozygous patients.
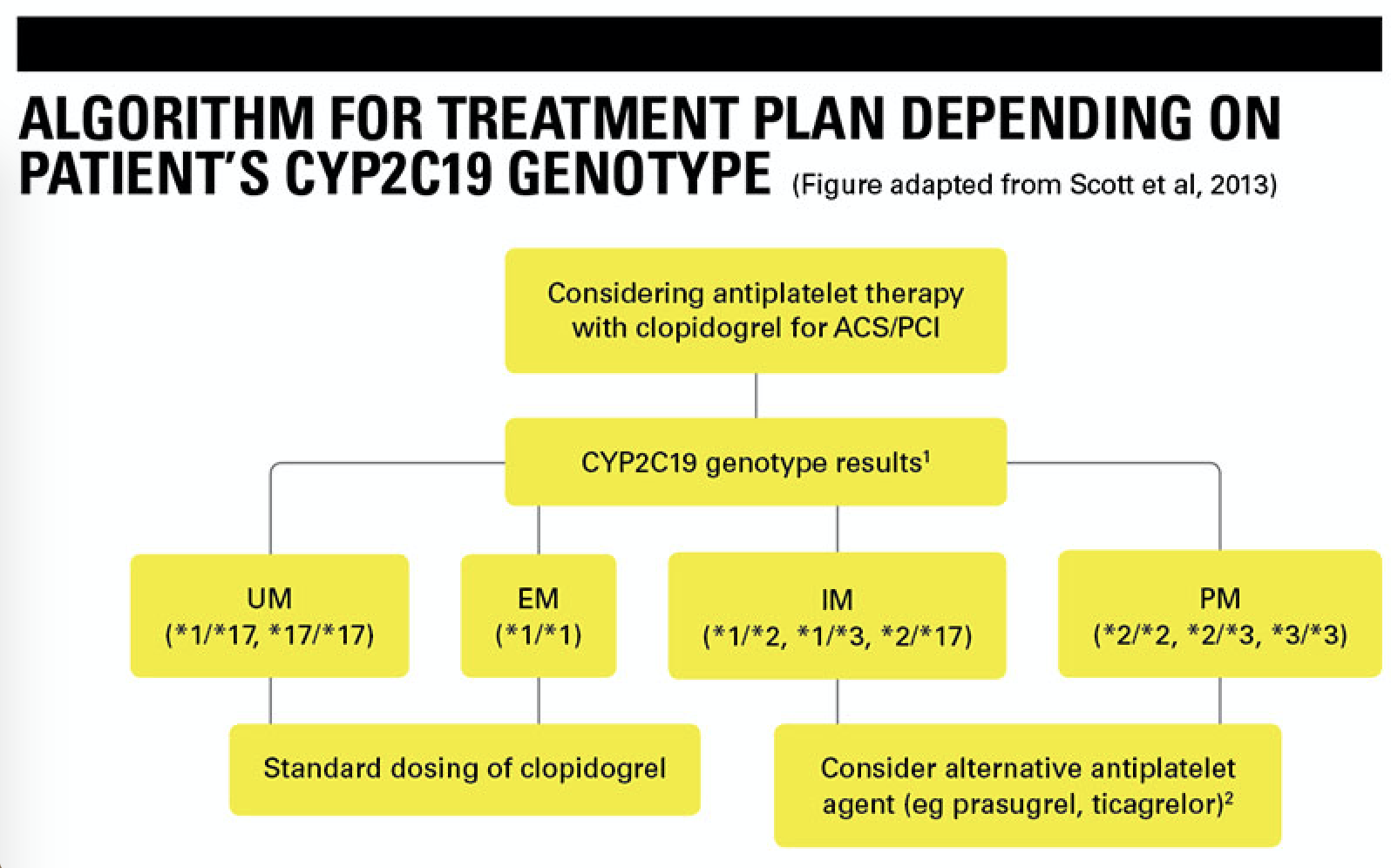
The figure below summarises the recommended algorithm for clinicians when planning a patient’s treatment course. In the clinical studies stated left, where some patients were subject to increased bleeding from clopidogrel, with genotyping data, it was evident that these were most likely individuals carrying *1 (wild type) or *17 (UM) allele, whereas those with *2-*8 alleles, a lower risk of bleeding would be observed, since they are poor metabolisers but a higher dose would be needed to confer any significant biological antiplatelet activity. For these reasons, the Clinical Pharmacogenetics Implementation Consortium (CPIC) guidelines on CYP2C19 genotyping for clopidogrel recommend alternative antiplatelet therapies, such as prasugrel, ticagrelor or indeed PAR-1 and αIIbβ3 inhibitors are considered when not clinically contraindicated. Although patients are genotyped, the benefits of prescribing clopidogrel should be carefully weighed against major adverse effects such as bleeding, in which case a potential antiplatelet benefit would be counterbalanced by the bleeding and other factors that contraindicate. For example, it has been reported that CYP2C19*17 (rs12248560) status confers significant enhanced clopidogrel metabolism but also increases risk of bleeding. Additionally, the presence of a novel CYP2C19 variant in a patient annotated as “probably damaging” or “pathogenic” without further investigations should be cautiously reported as its biological interaction with clopidogrel would be unknown. Likewise for variant identified as “benign” or “tolerated”, it should not be assumed, for example, to mimic the biological behaviour of known loss of function variants in response to clopidogrel. As for the IM phenotypes that have genotypes such as *1/*2 or *2/*17, it is recommended that alternative antiplatelet therapies are prescribed due to the high risk of adverse cardiovascular outcomes. Finally, clinicians should always remember that the aim of personalised medicine is ultimately to minimise disease risk and associated adverse effects.
Ibrahim Nakibingé is a Biomedical Scientist and IT Transition Analyst working in North West London Pathology.
Image credit | Science photo library
