Jenny Berryman, Hema Mistry and Paula Bolton-Maggs from the Serious Hazards of Transfusion (SHOT) scheme explain their latest annual report.

The Serious Hazards of Transfusion (SHOT) scheme has been running for 21 years now. It continues to collect and analyse anonymised information reported in the UK about serious adverse reactions and other serious adverse events (SAE) related to blood transfusion. The cumulative learning from past recommendations has led to the mitigation of many system faults that led to fatal and potentially fatal errors. Errors attributable to “human factors” persist, and so systems and practices must be re-assessed, and re-designed to minimise the effect of human error.
There are nine critical steps in the transfusion process from the point of request, sample taking, sample receipt, testing, component selection, component labelling, component collection, prescription and administration. Each step could potentially be performed by a different person and incorporates independent checks, which, if performed correctly, should detect the errors. The importance of working as a team is illustrated by cases reporting multiple errors at different points of these critical steps (see Case Study 1 below). SHOT recognises that incidents rarely result from a single point of failure and urges that at each step, staff do not assume that errors have not been made in previous steps but verify for themselves. Poor communication also contributes significantly to the occurrence of errors: Do not assume, verify.
The breakdown of all reports analysed and included in the Annual SHOT Report 2017 (published in July 2018, and available here ) is as shown in Figure 1. The number of preventable errors remains high, with 85.5% in 2017 compared with 87.0% in 2016.

Key laboratory messages
Knowledge and skills
- Laboratory staff should have an understanding of all component types, including their storage conditions, but most importantly their compatibility with the patient and specific requirements for certain patient groups e.g. gender, age, pregnancy and taking disease status into consideration.
- Laboratory staff are responsible for maintaining their own continuing professional development (CPD), including competency assessments.
- Laboratory staff must understand warning flags, know why they have appeared and acknowledge appropriately. Warning flags should never be overridden by laboratory staff without understanding the reason for them.
Shared responsibility and shared care
- Good communication is paramount between staff in the laboratory, between the laboratory and the clinical area and vice versa.
- It is important, when necessary, to look up, understand and maintain patients’ historical records and to seek out any further transfusion information that may be available for the patient from a shared care facility e.g. transplant, antibodies, adverse transfusion history. Never assume that something has been done: always double check.
Information technology
- Laboratories should have a contingency procedure for IT failure and perform a simulated situation competency which renders the laboratory information management systems (LIMS) out of action in order to test that the contingency procedures are robust.
- SHOT data continue to highlight that many errors are caused by overriding warning alerts. It is now time for LIMS suppliers to provide software that requires more than a keystroke to override the warning alert and meet transfusion guidelines.
Case study 1
A demographic data entry at sample entry results in a patient receiving ABO-incompatible fresh frozen plasma (FFP).
Five units of FFP were ordered by telephone for Patient 1. During the laboratory IT process, the copy and paste function was used to populate the sample identification number field. However, the sample ID number pasted into the sample ID field belonged to the previous patient (Patient 2).
At collection, the porter noted the discrepancy. The FFP was then re-labelled for Patient 2, but the biomedical scientist failed to note that the FFP was incompatible. The nurse administering the FFP noted the group was different to the patient but believed that group O components were compatible for all patients. This resulted in group O (Patient 2) FFP being administered to Patient 1 (group A).
There were four errors that occurred:
- Sample receipt – biomedical scientist took verbal telephone request but selected the wrong patient.
- Component selection – biomedical scientist did not identify that the wrong patient had been selected.
- Component labelling – biomedical scientist did not check label against the request. Porter detected the error, but the biomedical scientist re-labelled it for Patient 2 and did not notice the FFP was incompatible.
- Administration – nurse noted the group was different but believed that group O FFP was compatible for all patients.
Learning points
Group O FFP should only be issued for group O patients. Group AB is the universal group for FFP (but group A may be used, if group AB is not available).
Clinical staff are required to perform the critical bedside checks, including knowledge of compatibility, prior to administering the component. However, the laboratory staff must perform essential checks in the transfusion laboratory to ensure that the component is correct for the patient prior to it leaving the laboratory.
Deaths and major morbidity
There were 21 deaths where transfusion was implicated, and 112 additional cases where patients suffered major morbidity.
Twelve of 21 transfusion-related deaths reported in 2017 were due to pulmonary complications. An additional six were related to delays. Laboratory staff in particular should take note of the key SHOT message and laboratory recommendations to facilitate the rapid issue of blood components in emergency situations. Prompt initiation of “concessionary release” policies (enabling emergency issue of components that don’t meet best practice guidelines) and good communication are vital in emergencies.
Key recommendations
The very first SHOT report recommendation from 1997-8 states: “The bedside check is vital in preventing transfusion error. Staff should be vigilant in checking identification details of the component against those of the patient.” The recommendations from the 2017 SHOT report reflect the persistence of human factors and the roles of the correct application of knowledge, supported by effective use of IT in reducing transfusion error:
1. Knowledge and skills: Training in ABO and D blood group principles is essential for all laboratory and clinical staff with any responsibility for the transfusion process. This should form part of the competency assessments.
2. Information technology: All available information technology (IT) systems to support transfusion practice should be considered and these systems implemented to their full functionality. Electronic blood management systems should be considered in all clinical settings where transfusion takes place. This is no longer an innovative approach to safe transfusion practice, it is the standard that all should aim for.
3. TACO: A formal pre-transfusion risk assessment for transfusion-associated circulatory overload (TACO) should be undertaken whenever possible, as TACO is the most commonly reported cause of transfusion-related mortality and major morbidity.
Patients who develop respiratory distress during or up to 24 hours after transfusion where transfusion is suspected to be the cause must be reported to SHOT. The national comparative audit of TACO in 2017 demonstrated that risk factors are being missed.
Key SHOT messages
1. Guidelines or rules? Guidelines must not be translated into inflexible rules. Proportionate application of knowledge and experience may lead to a different course of action in individual circumstances. But the final bedside check is a rule and must be completed in full.
2. Basic training: It is essential that all staff participating in transfusion fully understand ABO groups so that they can recognise potential ABO-incompatibility.
3. IT systems have the potential to increase transfusion safety by minimising human factors and should be considered for all transfusion steps.
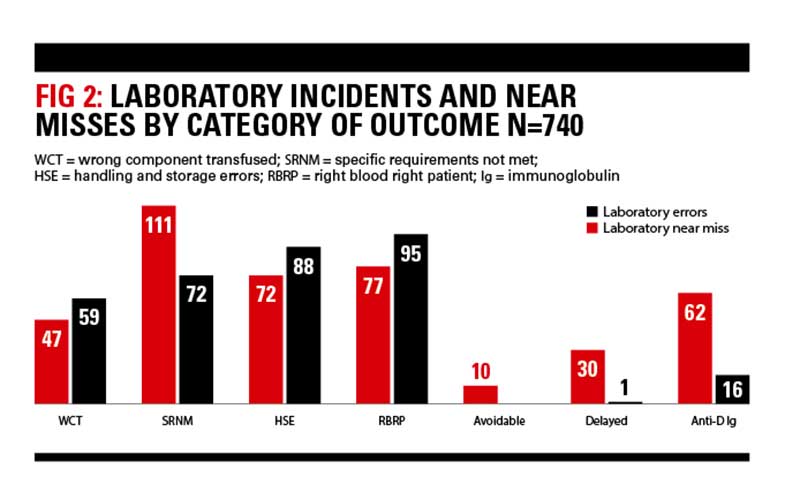
Laboratory errors
There were 740 errors reported to SHOT in 2017 that originated from the laboratory comprising 409 errors where the patient was transfused, and 331 near misses.
ABO-incompatible transfusions
There were four ABO-incompatible FFP transfusions due to errors at sample receipt (1), case 1, testing (1) and component selection (2) and 1 ABO-incompatible platelet transfusion due to a component selection error. Two other ABO-incompatible transfusions occurred as a result of clinical errors (red cells due to administration error and platelets due to a wrong blood in tube error).
Improving safety
The relative risks of transfusion today are low. In order to further improve transfusion safety, laboratory staff (and all staff involved in the transfusion process) should take heed of the key SHOT messages and recommendations. Good communication, full understanding of transfusion principles, diligent checking and re-checking, all supported by effective use of IT solutions will help to mitigate the risk of transfusion error.
Jenny Berryman is a Specialist Advisory Panel Representative (Transfusion)
Hema Mistry is a Laboratory Incidents Specialist
Paula HB Bolton-Maggs is Medical Director, all at SHOT.
Image credit | Science Photo Library




