This month's top tech news stories

Immunovia
Pancreatic cancer
Immunovia has received final approval to begin patient testing for the IMMray PanCan-d test – the first blood test on the market dedicated to the early detection of pancreatic cancer.
Approval was received from the Massachusetts Department of Public Health on 3 August.
The test recognises biomarker signatures and has the potential to increase the survival of patients by detecting pancreatic cancer when surgical resection is possible.

Radiochemistry
Medicines Discovery Catapult has relaunched radiochemistry at the Wolfson Molecular Imaging Centre in Manchester.
The facility, which was closed in 2020, includes the multi-million pound Cyclotron – one of only a handful in the UK.
It will supply hard-to-make radiochemicals to drug discovery biotechs and academic innovators – increasing the UK’s potential to discover new and better therapies for patients, faster.
BD
Test for variants
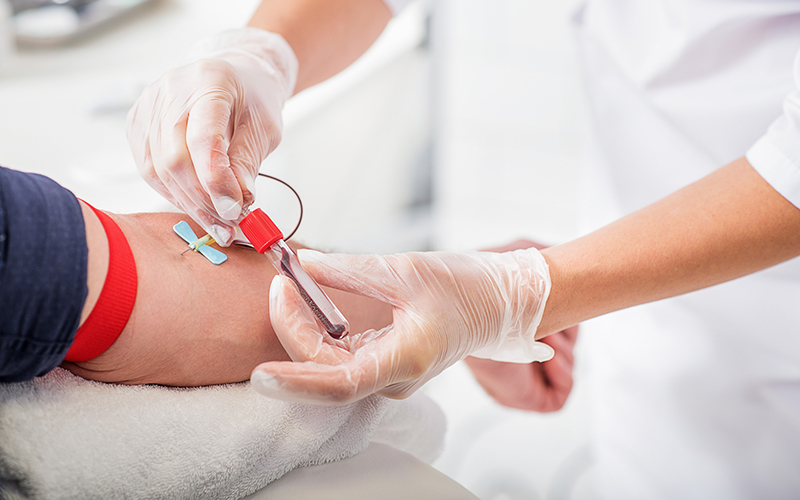
BD and CerTest Biotec have announced the CE Mark for a molecular test to detect and identify certain SARS-CoV-2 variants.
VIASURE SARS-CoV-2 Variant Real Time PCR Detection Kit for BD MAX can be used as a combined test with VIASURE SARS-CoV-2 (N1+N2) Real Time PCR Detection Kit for BD MAX, or as a reflex test to run variant identification on a SARS-CoV-2-positive sample.
The test snaps into the test-specific position on the BD MAX ExK TNA extraction strip, supplied by BD.
Image credit | Frédéric-Bergeron |Chris-Bull-Alamy| iStock
